May / June Issue of Vein Specialist
Venous Disease and Pregnancy: Navigating a Difficult Course

Maggie Arnold, MD, FACS
Associate Professor of Surgery
MedStar Health
Baltimore, MD

Qingwen Kawaji, MD
Chief Resident, General Surgery
MedStar Health
Baltimore, MD
Pregnancy increases women’s risk for lower extremity venous disease, including venous insufficiency and venous thromboembolism (VTE). This elevated risk affects both pregnancies and the immediate postpartum period (3 months after delivery). Recent reports showed an incidence rate of 0.67 VTE cases per 1000 pregnancies (0.071%) during pregnancy and 0.045 per 1000 pregnancies (0.004%) during the postpartum period (1). Physiological changes of the lower extremity venous system during pregnancy are responsible for this elevated risk; these changes include increased cardiac output, decreased systemic vascular resistance, increased venous capacitance, mechanical compression of the uterus on central venous systems, and hypercoagulable state (2).
Currently, no data support the routine use of VTE prophylaxis in the general pregnant population except for women who have inherited hypercoagulable disease. Diagnosis and management of VTE have unique challenges and should not follow the typical pathway of a non-pregnant person. Venous duplex ultrasound remains the initial preferred study of choice, with follow-up magnetic resonance angiography if iliac or inferior vena cava thrombosis is suspected. CT scans should be avoided during pregnancy, but can be safely used postpartum.
Medical management of VTE during pregnancy includes low molecular weight heparin (LMWH) and unfractionated heparin if LMWH is contraindicated. In the situation of heparin-induced thrombocytopenia, fondaparinux, argatroban, and danaparoid can be used. Warfarin should be avoided during pregnancy because it is a teratogen and crosses the placenta. Direct oral anticoagulants, such as dabigatran or apixaban, should also be avoided due to limited data on fetal safety.
Invasive therapies for treating VTE during pregnancy are reserved for patients with significant symptoms and, when performed, should be approached from a multi-disciplinary perspective with coordination with obstetricians and experts in other maternal-fetal medicine domains. Several options exist for treating extensive and symptomatic iliofemoral VTE: catheter-directed thrombolysis (CDT), pharmacomechanical thrombectomy (PMT), and operative thrombectomy. Local thrombolytic therapy can effectively treat extensive iliofemoral VTE while minimizing the risk of bleeding complications. Although a risk of bleeding remains, local thrombolytic therapy is acceptable given the clinical disease severity. The main concern for catheter-directed therapy is the radiation exposure required for the procedure, although there are reports on contrast-free and radiation-free catheter placement using ultrasound only (3). Moreover, with intravascular ultrasound, radiation exposure can be further reduced. Limited studies report on the outcomes of these invasive procedures. However, some suggest that CDT alone is less effective than with PMT in terms of thrombectomy efficacy and incidence of post-thrombotic syndrome (4). Although some studies have described using percutaneous thrombectomy as a stand-alone approach for treating iliofemoral VTE, the literature surrounding pregnancy is scarce. Stenting should be avoided in pregnancy as compression of the venous system with an enlarging uterus may reduce its patency.
Minimal literature exists regarding iliofemoral stenting during pregnancy, although a recent paper by Villalba et al. showed continued patency of dedicated venous stents despite pregnancy (5).
In conclusion, treatment of VTE during pregnancy remains a scarcely studied topic that would benefit from further investigations for indication and optimal treatment.
References
- Ram S, Ram HS, Neuhof B, Shperling RB, Chodick G, Yogev Y. Venous thromboembolism during pregnancy: Trends, incidence, and risk patterns in a large cohort population. International Journal of Gynecology & Obstetrics. 2023;160(3):962-968. doi:10.1002/ijgo.14414
- Taylor J, Hicks CW, Heller JA. The hemodynamic effects of pregnancy on the lower extremity venous system. Journal of Vascular Surgery: Venous and Lymphatic Disorders. 2018;6(2):246-255. doi:10.1016/j.jvsv.2017.08.001
- Huegel U, Surbek D, Mosimann B, Kucher N. Radiation- and contrast medium-free catheter-directed thrombolysis for early pregnancy-related massive iliocaval deep venous thrombosis. J Vasc Surg Venous Lymphat Disord. 2019;7(1):122-125. doi:10.1016/j.jvsv.2018.06.007
- Lu ZX, Wei HL, Shi Y, Huang H, Su H, Chen L. Safety and Efficacy of Endovascular Treatment on Pregnancy-Related Iliofemoral Deep Vein Thrombosis. Clin Appl Thromb Hemost. 2022;28:10760296221124904. doi:10.1177/10760296221124903
- Villalba L, Larkin TA. Outcomes of dedicated iliac venous stents during pregnancy and postpartum. J Vasc Surg Venous Lymphat Disord. Published online April 6, 2023:S2213-333X(23)00137-3. doi:10.1016/j.jvsv.2023.03.009

Venous Stasis Skin Change Mimickers

Eri Fukaya MD, PhD
Division of Vascular Surgery, Vascular Medicine Section
Stanford University School of Medicine
Palo Alto, CA
It is not uncommon to have patients referred for evaluation of “venous stasis changes in the gaiter area.” While these may be of true venous etiology, there are mimickers we should look out for.
Venous Stasis Skin Changes
Alterations in venous tone and shear impair nitric oxide production and promote the release of vasoactive agents and inflammatory cells and cytokines. This promotes leukocyte recruitment, rolling, adhesion, and transmigration through the endothelium of the vein wall triggering an inflammatory response. Activated endothelial and inflammatory cells can degrade components of the extracellular matrix, including collagen, elastin, laminin, and fibronectin, by releasing oxygen free radicals and MMPs. The increased expression of MMPs and other proteinases by the vessel wall breaks down the vascular extracellular matrix, leading to increased vascular permeability. This allows for erythrocyte extravasation, which releases ferritin and ferric ion and causes hemosiderin deposits in the skin resulting in hyperpigmentation.
Another commonly seen skin change is lipodermatosclerosis. This localized, hardened skin occurs due to sclerosing panniculitis. It is thought that perivascular inflammation results in panniculitis. Histologically, there is initially a thickening of the pannicular septa, a mild inflammatory infiltrate, and early adipocyte necrosis toward the center of the fat lobule. As it progresses, the septa become increasingly fibrotic, with fibrous strands extending into the fat lobule.
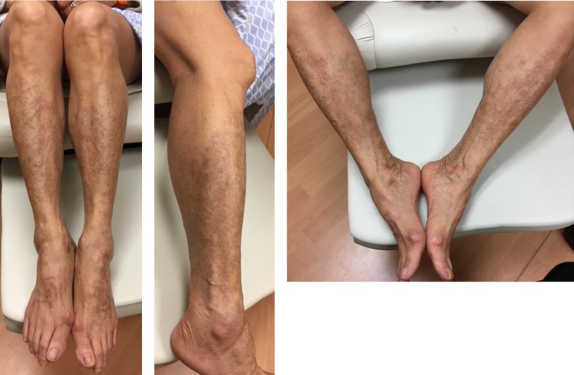
Hyperpigmentation mimicker: Pigmented purpura, aka capillaritis, aka Schamberg’s disease
Pigmented purpura is characterized by petechiae and bronze discoloration of the skin on the lower extremities involving the pretibial region and extending proximally (Figure 1). It predominates in men in their 50s but can occur in all age groups and sex. This is believed to be due to a cutaneous hypersensitivity reaction that causes capillary fragility and permeability, leading to erythrocyte extravasation and hemosiderin deposits noted on biopsy. Histopathologically, extravasation of erythrocytes with hemosiderin deposition, a perivascular lymphocytic infiltrate centered on the superficial capillaries, and endothelial cell swelling are seen. Patients are asymptomatic and the clinical course is benign.
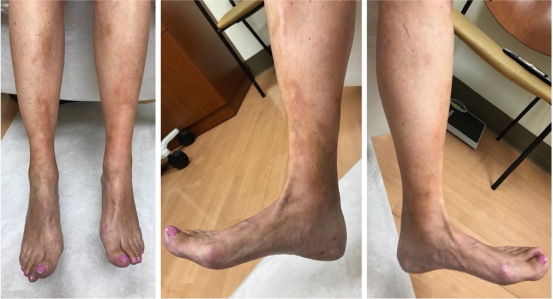
Lipodermatosclerosis mimicker: Morphea
Morphea, also known as localized scleroderma, is a condition that causes hardening and discoloration of the skin and is an autoimmune disorder (Figure 2). Histologically, perivascular infiltrate predominantly composed of lymphocytes and plasma cells, with some eosinophils and macrophages, is seen. It presents as erythematous skin lesions with some itching and tenderness. The lesions are usually self-limiting, but some patients can develop ongoing symptoms.
Lipodermatosclerosis mimicker: Erythema nodosum
Erythema nodosum is an acute nodular septal panniculitis, characterized by the sudden onset of erythematous, firm, solid, deep nodules or plaques painful on palpation and mainly localized on extensor surfaces of the legs. These nodules have a histological appearance marked by acute inflammation of the dermo-hypodermic junction and interlobular septa of the hypodermic fat, evolving without necrosis or sequelae. Its cause is unknown but can be a sign of some other infection, disease, or of sensitivity to a drug. These bumps can be tender, painful, and warm to the touch.
Figure 1: Pigmented purpura.
Figure 2: Morphea in a patient with diffuse cutaneous scleroderma and Raynaud phenomenon.