Table of Contents
Message from the Editor: What Room?
Steve Elias, MD
Membership Matters
VENOUS2023 Introduction
Mikel Sadek, MD
Reflections and Advice from the 2022 AVF Grant Winners
Andrea Obi, MD
Venous Imaging
Ascending and Descending Venography
Amber Liles, MD, MPH
Imaging for Lymphatic Disease
Brett Carroll, MD
IVUS: What is new?
David Chalyan, M.D., M.Sc. and Denysse Castañeda
CT/MR Venography
Natalie Sridharan, MD
Major Concerns About Venous Imaging in the Routine Clinical Practice
Nicos Labropoulos, PhD
The Future of Novel Thermal Imaging in Venous Disease
Ariel Soffer, MD, FACC, Shayna N. Soffer, David Wright, MB, FRCS, Sarah Melvin, MSPH
International
AVF Abroad & An Update from EVF
Kathleen Ozsvath, MD
Working on International Cooperation Matters
Tomasz Urbanek, MD
American Venous Forum and Latin America United
Alvaro Orrego, MD
Appropriateness: EVF Congress 2022 in Venice, Italy
Dominik Heim, MD
Copyright © 2022 by The American Venous Forum. All rights reserved.
EDITOR-IN-CHIEF
Steve Elias, MD
EXECUTIVE EDITOR
John Forbes, MBA
MANAGING EDITOR
Allie Woodward
PUBLICATION DESIGNER
Christine Rataj
Message from the Editor: What Room?

Steve Elias, MD
Editor-in-Chief, AVF Newsletter
Communications Councilor, AVF Board of Directors
When you feel the urge and you need to go, you have choices. But what to choose? Maybe the semantics or euphemisms confuse you. We have done a bad job. The myriad of names for the proper place to relieve oneself are confusing. Let’s go through a few and rate the monikers 1-10:
1. Bathroom
Clearly one can bathe in the bathroom in a bath. But many bathrooms in public places or restaurants don’t have a bath. There is usually a toilet and a sink. So why when it is that time of urgency do we go to the bathroom? You don’t want a bath. Rating: 5
2. Comfort Station
This may actually make sense. We’ve all experienced comfort after using the Comfort Station. So, I kind of get it. But the word comfort is somewhat misleading. Someone may think the comfort station is used for other types of comfort such as emotional and enter thinking that’s what they’re going to get. But as a name for what it’s supposed to be used for, not bad. Rating: 8
3. Washroom
Similar to the bathroom. A lot of euphemistic washing. One can usually wash in the washroom. There is almost always a sink in addition to the necessary accoutrement, the toilet. But the name implies that the main intent is to wash. To be clear, it is not. Rating: 6
4. WC (Water Closet)
This is the European connection. The WC implies that water is stored in this closet. Upon opening the door, one would expect to see cases of bottled water and may enter if thirsty. There are no water bottles in most water closets. But there is water in the toilets and sinks…not to drink. Rating: 4
5. Restroom
While this is best understood as the euphemism for needing to relieve yourself, it is still imprecise. Does anyone go to the restroom to rest? Of course not, we go to the restroom to do some important work, not rest. But in any case, this is the most used term. Rating: 9
So while imaging options for venous and lymphatic disease are established and evolving and our future is together when researching the past the name Thomas Crapper inevitably comes up when speaking about bathrooms. He was an English plumber and businessman born in 1836 who founded the plumbing equipment company, Thomas Crapper and Company. He was an inventor who held 9 patents related to waste management. Urban folklore has promulgated the myth that the term for bowel relief comes from his name. Untrue. Most likely it came from Middle English origin and the Dutch word “krappen”, to pluck off and the Old French ”crappe”: siftings, waste or rejected matter. Thomas Crapper had the misfortune or fortune of becoming a plumber with the surname Crapper. Famous or infamous? You decide. And in this issue of Vein Specialist you can decide when you need what type of imaging to help patients. The better to see you with. Because if you can’t see it, you can’t treat it and you can’t help patients. Please read this issue of Vein Specialist whether at home, in the restroom, bathroom, WC, washroom or comfort station. No matter where, you’ll be able to better use the right imaging for the right patient. And when you flush and it all disappears, thank Thomas Crapper, the inventor of the toilet. Not the inventor of the euphemism.
Free Upcoming Webinar
September 13, 2022 — 8:00 – 9:15 PM ET
Venous Stenting with Venovo: One and Done?
Iliofemoral Stenting & You: Tips and Tricks for Success in the Deep Venous Space – Sheila Blumberg, MD
Now or Later? Should We Place a Venovo Stent Following Acute DVT Thrombectomy with Aspirex? – Patrick Muck, MD
The Great Divide: To Cross or Not Cross the Inguinal Ligament with Dedicated Venous Stents? – Erin Murphy, MD
Back by Popular Demand! Venovo and its role in Complex Iliofemoral Reconstructions – Kush Desai, MD
Supported by an education grant from:

VENOUS2023: One World, Many Voices Our Future is Together

Mikel Sadek, MD
Chair, AVF Annual Program Committee
The Program Committee for VENOUS2023 has the vision to bring you the most comprehensive, compelling, and cutting-edge venous program to date. We had the humbling objective of manifesting this concept for the world of venous and lymphatic disease to best serve the American Venous Forum and its membership.
Our group deliberated how best to capture the vision for the upcoming annual AVF meeting. We carefully reviewed the feedback from last year’s attendees and shared our collective experience as members of the Program Committee. An animated discussion ensued.
Today, we are facing countless challenges and each one of us has been touched by events in our one world. Each one of us has a unique perspective based on our cumulative life experience. Each one of us has something to say, and we speak with many unique voices. Little is guaranteed in life except for death and taxes, as per Benjamin Franklin. Certainly, time is a constant, and with the known passage of time, we take stock of the present and look to the future. Sometimes we like what we see, and sometimes we don’t. Regardless, we push together even to Arthur C. Clarke’s Childhood’s End, or for us, to San Antonio, Texas, for VENOUS2023.
To that end, the theme for VENOUS2023 is:
One World, Many Voices: Our Future is Together
Clinical and Scientific Voices
First and foremost, the goal is to bring the highest quality clinical and basic science from all corners of the earth—and a sphere has many corners. The venous world has many voices including lymphatic, aesthetic, deep, superficial, thrombus…to name a few. Moreover, there will be an increased effort to incorporate expert clinician experience to augment the discussions on basic science, retrospective reviews, and randomized controlled trials.


International Voices
We are expanding on prior initiatives to incorporate an additional and more robust International Session. With the assistance of our industry partners, a novel International Industry Session will be presented, which has the potential for tremendous future expansion.
All Voices
We will be augmenting the Diversity, Equity, and Inclusion aspects to the program. We understand that this will always be a work in progress, but we aim to have a program that is closer to being representative of the population that we treat. This includes, but is not limited to, the program content, presenters, keynote presenters, moderators, and the audience.
We hope to bring these exciting aspects – and many others – to VENOUS2023 in San Antonio, TX, to build on the prior meeting successes, and to continue to lay the path for future growth. The vision is to become the central repository for the many voices of those involved in venous and lymphatic disease, because the way to move forward, for our patients and ourselves, is together.

Reflections and Advice from the 2022 AVF Grant Winners

Andrea Obi, MD
Chair, AVF Research Committee
Past Chair, AVF Digital Media Committee
Member, AVF Newsletter Committee
2022 Boston Scientific Research Grant Recipient
Where did you get your inspiration for this project?
I think a lot of research ideas come from clinical observations. For me, evaluation of chronic venous disease was a frequent consult during my clinical rotations. Venous disease restricts daily activities, is costly to the healthcare system, and requires extensive outpatient resources.
How far in advance did you start writing / working on the project?
My mentor Dr. Sachdev has been fantastic in bringing me up to speed with the data and results that were already established when I first began my dedicated research time. This gave me the opportunity to work on tying everything together and solidifying our specific research aims for the project a few months before the grant deadline.
What was the most challenging part of putting together the proposal?
The most challenging part of the proposal for me was to appropriately convey our ideas in a way that was easy to understand and follow, all while highlighting the translational aspects of it. The process of taking an idea from either ends of the spectrum and build upon it enough to form a clear and concise proposal was the most difficult to grasp.
What advice would you give to someone writing a Jobst or Boston Scientific grant this year?
It’s very important and valuable to be able to jot down all the ideas that come into mind and refer to them later on because you never know where and how they might fit into your proposal. I find the speech-to-text dictation functions on phones to be useful if I’m on a long drive; other times, I’ll simply type up what comes across my mind on the notepad app of my phone.
What was something unexpected that you encountered in the writing or submission process?
Budgeting was something that I did not have a lot of experience with; the process of appropriately allocating funds to each aspect of a project was a learning process. It really put in perspective the detailed planning that goes into setting up a project.
2022 Jobst Grant Recipient
Where did you get your inspiration for this project?
I was fortunate to receive a Marie Sklodowska Curie Research Fellowship in Galway, Ireland, during my residency. I shared an office with a postdoctoral researcher, Andrew Cameron, who recognized that there were numerous unmet needs in management of venous leg ulcers and was awarded a commercialization grant to develop technologies that improve the delivery of compression therapy. We experimented with a few different approaches to monitor pressure after a dressing was applied, but the most promising strategy involved the use of a remote pressure sensor that sits underneath a wrap or bandage.
How far in advance did you start writing / working on the project?
Technically, I started writing the grant about a month before it was due, but I had been working out a structure for the study for years prior.
What was the most challenging part of putting together the proposal?
The real challenge for me was the timing of the due date, which was approximately 1 month after I started my first faculty position. I had just moved, started my first job, was learning how to be an attending for the first time, had to learn a new hospital and prepare for the boards, and then had to write and submit a grant at a brand-new institution!
What advice would you give to someone writing a Jobst or Boston Scientific grant this year?
It’s all about the mentors. I started a new job where there really was no active venous research program or clinical practice. However, I have two amazing clinical partners/mentors who were able to help me put together a research plan that fits within our infrastructure and will hopefully allow us to answer some interesting questions. I also reached out to some of my mentors from training.
What was something unexpected that you encountered in the writing or submission process?
I had never written a grant that involved a clinical trial, so developing the study protocols has been a completely new experience. As I put together the grant various questions arose that I needed to address: how long it takes for a patient to complete a survey? Where do you put the patient if they come for an office visit, and you need to run tests? Fortunately, my mentors and our clinical coordinator team have run a number of clinical trials before and were able to steer me in the right direction.
Now Accepting Research Grant Submissions
BSCI Translational Research Grant
Deadline Approaching — August 15, 2022
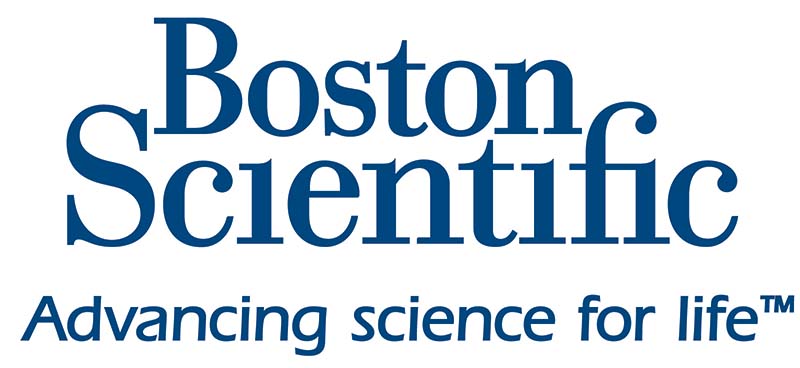
$85k over a period of 2 years for an original, translational research project in venous disease addressing one or more of the following:
- Nonthrombotic venous obstruction
- Venous thromboembolism
- Superficial venous insufficiency
International submissions accepted!
Submission Deadline: Monday, August 15, 2022
Jobst Research Grant

$85k over a period of 2 years for clinical research in venous diseases, lymphatic diseases, or lipedema with an emphasis on:
- Prevention of disease and its progression
- Diagnosis of disease
- The science of management of the above conditions, especially with compression therapy
Submission Deadline: Monday, October 15, 2022
Ascending and Descending Venography: Adjunct Tool for Evaluation of Pelvic Venous Disorders and Deep Venous Disease

Amber Liles, MD, MPH
Interventional Radiology, University of Michigan
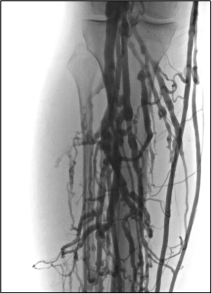
Noninvasive imaging studies such as venous duplex and CT venogram (CTV) are a mainstay in the assessment of pelvic venous disorders but may suggest multiple contributing etiologies for the patient’s symptoms, making treatment decisions challenging. For example, consider a 32-year-old female with multiple prior pregnancies presenting to clinic with asymmetric left lower extremity swelling, pelvic pain that is worse during menstruation, and painful vulvar varicosities with dyspareunia. CTV shows an enlarged left gonadal vein, bilateral para-uterine varicosities, compression of the left common iliac vein (LCIV) by the right common iliac artery, and vulvar varicosities without a clear source. This is not an uncommon scenario, and it is not always clear what potential pathology should be targeted for treatment. Should the patient undergo gonadal vein embolization, LCIV stenting, both, or neither? In these challenging cases, descending venography can be a helpful adjunct to noninvasive studies, allowing for more complete flow assessment of the deep veins of the pelvis.
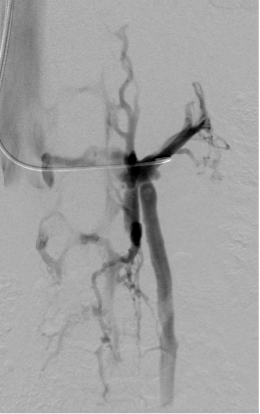
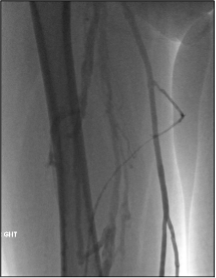
For a comprehensive evaluation of pelvic venous disorders, we often start with left renal venography. A catheter is advanced in the left renal vein near the hilum, and venography is performed with Valsalva maneuver, assessing for both gonadal vein reflux and signs of renal vein compression (Figure 1). If there is concern for Nutcracker syndrome, evaluation of the left renal vein with intravascular ultrasound (IVUS) and manometry is performed. If the patient has gonadal vein reflux and renal vein compression without Nutcracker syndrome, repeat left renal venography and manometry with an occlusion balloon in the left gonadal vein can better estimate the effect of potential left gonadal vein embolization on renal vein flow.
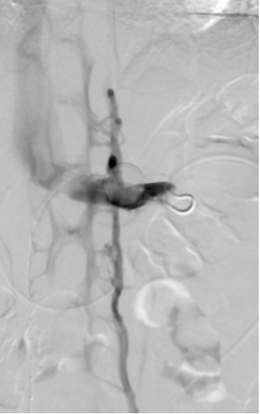
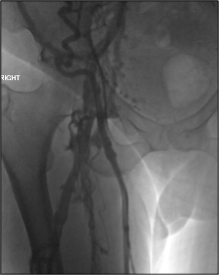
The catheter is then advanced into the left gonadal vein and reflux into deep pelvic veins is assessed. If a tilt-table is available, reverse Trendelenburg positioning can simulate a more physiologic state and is preferred. The same is repeated for the right gonadal vein, usually originating directly from the inferior vena cava (Figure 2).
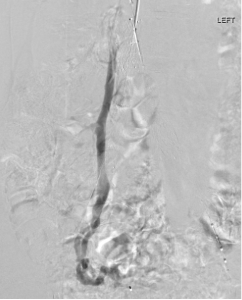
If gonadal vein reflux does not fully explain pelvic symptoms, further evaluation of the iliac veins is performed during the same procedure. The LCIV is assessed for external compression first with venography and then with IVUS if venographic signs of compression exist (Figure 3). Both internal iliac veins (IIV) are evaluated, assessing for reflux and cross-pelvic collateralization. In patients with perineal or gluteal varicosities, potential pelvic escape points may not be immediately apparent on IIV injection but may be more readily seen with a balloon occlusion catheter in the IIV. Additionally, direct injection of the greater saphenous veins to evaluate for an external pudendal contribution to varicosities can be helpful in select patients.
After either targeted or comprehensive assessment with descending venography, appropriate endovascular treatment can be rendered, often during the same procedure. In some patients, multiple underlying etiologies for a pelvic venous disorder may still be evident. In these cases, a multidisciplinary approach with potential staging of treatment is recommended
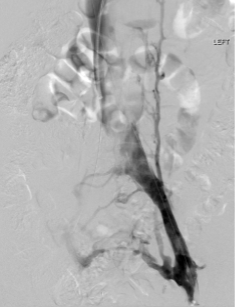
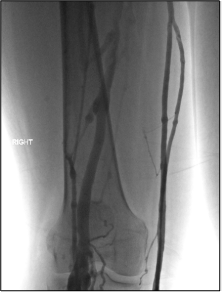
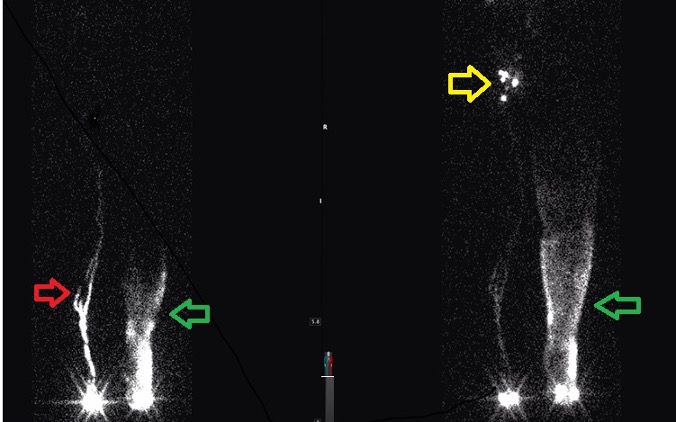
Similar to descending venography, ascending venography can be a helpful adjunct in the assessment of iliofemoral or femoropopliteal deep venous disease. Venous duplex studies, as currently performed in most practices, generally show vessel patency and presence of chronic thrombus well but provide limited assessment of global flow and usually no evaluation of the deep femoral vein. Consider this scenario: You’re performing iliocaval recanalization in a patient with IVC filter-associated iliocaval thrombosis. A duplex study shows chronic thrombus in the recanalized femoral veins. Three hours into the procedure, you’ve successfully recanalized the iliocaval segments but you realize that there is severe bilateral femoral and deep femoral disease, limiting inflow to the pelvis. In a patient with femoral disease on a preprocedural duplex study, ascending venogram provides additional information about severity of disease and flow dynamics between the superficial and deep system, allowing for better procedural planning and patient counseling.
At our institution, ascending venography is performed with injection of contrast into a small vein of the foot on a tilt-table with the patient in near standing position. Compression is often applied to the superficial venous system below the knee to allow for better filling of the deep veins. The evaluation includes assessment of not only the femoropopliteal segment but also assessment of the tibial veins, the deep femoral vein, and the greater saphenous vein. In practice, there can be a fair amount of discrepancy between venous duplex studies and ascending venograms regarding vessel patency; therefore, it’s important to know the strengths and weakness of both and use them as complimentary tools in appropriate patients.
Click images below to enlarge.
Figure 1. Left renal venography in two different patients. A, left renal venography shows reflux into a dilated left gonadal vein (arrow) and filling of the lumbar venous plexus (arrowhead). B, left renal venography shows flattening of the vein centrally with collateral flow through the lumbar plexus (arrowheads) but no reflux into the left gonadal vein.
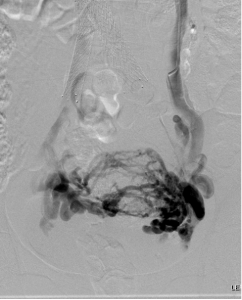
Figure 2. Descending gonadal venography. A, right gonadal venography shows reflux of contrast into right para-uterine veins. B, left gonadal venography shows reflux of contrast into large pelvic varicosities with cross-pelvic collateralization to the right internal iliac vein. This patient went on to have bilateral gonadal vein sclerosis and embolization.
Figure 3. Left iliac venography shows extrinsic compression of the central left common iliac vein with left internal iliac vein reflux, cross-pelvic collateralization, and filling of an ascending lumbar vein.
Figure 4. Ascending venogram of the right lower extremity. A, normal filling of deep and superficial veins of the calf with eventual drainage into the normal popliteal vein. B, normal appearance of the popliteal and peripheral femoral veins (arrow). C, postthrombotic change of the femoral vein (black arrow) with preferential flow to the deep femoral (white arrow) and greater saphenous veins (block arrow). D, the right iliac veins are occluded.
Imaging for Lymphatic Disease

Brett Carroll, MD
Director, Vascular Medicine
Co-Director, Raynaud’s Center, Beth Israel Deaconess Medical Center
Options for the evaluation of the lymphatic system are increasing. Objective assessment of lymphatic function can be clinically useful, particularly for patients with unexplained limb edema. Classic physical exam findings (eg, Stemmer’s sign, distribution of edema, and certain skin changes) are very consistent with lymphedema. In cases with a clear prior lymphatic insult, such as regional lymphadenectomy or radiation, there is generally little need to perform lymphatic imaging to confirm the diagnosis. However, in patients with limb swelling without clear etiology on history or exam, additional lymphatic imaging can be helpful to objectively evaluate for the presence of lymphatic dysfunction.
Invasive lymphangiography, with direct cannulation of a lymphatic vessel or lymph node, had been the traditional modality for lymphatic assessment. Though features of slow transit or obstructive lymphatic anatomy can be visualized with invasive lymphangiography, it is now more commonly utilized in the cases of central lymphatic disease such as a chylothorax or chylous ascites. Nuclear lymphoscintigraphy has surpassed invasive lymphangiography as the current preferred modality for evaluation of lymphatic function at most centers (1). Protocols vary by institution. Injections of sulfur colloid bound to a nuclear tracer (most often technetium-99) are performed in the distal feet or hands, and the tracer is taken up by the lymphatic system. Transit of the radiotracer is serially imaged over 2 to 4 hours. Delayed or decreased uptake of the tracer in the inguinal or axillary lymph nodes is consistent with lymphatic dysfunction. The presence of channels can also be evaluated. Additional findings like dermal backflow are also consistent with lymphatic congestion and dysfunction. Nuclear lymphoscintigraphy offers high specificity and sensitivity. It has few adverse effects – there are rare allergic reactions and the injections can be transiently uncomfortable. Nuclear lymphoscintigraphy is an excellent modality to evaluate for overall lymphatic function, but lacks detailed anatomic evaluation limited by the granularity of nuclear imaging. Interestingly, there is overlap between venous and lymphatic disease with older nuclear lymphoscintigraphy studies demonstrating worsened lymphatic function in concordance with more severe chronic venous disease. Understanding this interplay is important when placing the findings of lymphoscintigraphy in the clinical context of patients with concomitant venous disease (2).
For a more specific evaluation of localized lymphatic dysfunction and anatomy, utilization of indocyanine green (ICG) lymphangiography is growing. ICG is a fluorescent dye that is injected intradermally in the hand or foot. It has rapid transit time within the lymphatics and allows for real time imaging of superficial lymphatic flow and anatomy. ICG maybe more sensitive than nuclear lymphoscintigraphy to detect early-stage disease (3). Additionally, there is clearer anatomic visualization of the superficial lymphatics and localized areas of dysfunction in a variety of patterns and severity. Such findings can be useful to guide both manual lymphatic drainage and target specific disease locations for lympho-venous bypass surgeries. ICG lymphangiography is generally only available at high-volume lymphatic centers.
Finally, there is growing utilization of magnetic resonance imaging (MRI) to evaluate lymphatic anatomy and the sequalae of the disease. MR lymphangiography involves injection of gadolinium intradermally and can demonstrate detailed lymphatic anatomy – how best to utilize such findings is still evolving (4). MRI is also valuable in evaluating the composition of the affected limb. Lymphedema leads to adipose tissue deposition and MRI offers the ability to differentiate fat from edema in the limb. Such an evaluation is instrumental in assessing appropriate candidates for liposuction and localizing the areas of adipose deposition to guide the surgical approach (5).
Depending on the clinical presentation, the evaluation of lymphedema may not require any imaging at all. However, in patients with unclear etiology to limb swelling or patients undergoing consideration for lymphatic surgery, a multimodality imaging assessment can be incredibly helpful in the diagnosis and management of lymphatic disease.
Figure 1. Nuclear lymphoscintogram in a patient with left lower extremity lymphedema. Left panel is image captured at 30 minutes demonstrating a lymphatic channel in the right lower extremity (red arrow) and dermal backflow (green arrow). The right panel is image captured at 2 hours with good uptake in the right inguinal lymph nodes (yellow arrow) with continued slow transit time and persistent dermal backflow in the left lower extremity.
References:
- International Society of Lymphology The diagnosis and treatment of peripheral lymphedema: 2013 consensus document of the International Society of Lymphology. Lymphology. 2013; 46:1–11.
- Bull RH, Gane JN, Evans JE, Joseph AE, Mortimer PS. Abnormal lymph drainage in patients with chronic venous leg ulcers. J Am Acad Dermatol. 1993; 28:585-90.
- Mihara M, Hara H, Araki J, et al. Indocyanine green (ICG) lymphography is superior to lymphoscintigraphy for diagnostic imaging of early lymphedema of the upper limbs. (2012) PloS one. 7 (6): e38182.
- Polomska AK, et al. Imaging technology of the lymphatic system. Adv Drug Delivery Reviews. 2021;170:294-311.
- Granoff MD, Johnson AR , Shillue K, et al. A single institution multi-disciplinary approach to power-assisted liposuction for the management of lymphedema. Ann Surg. 2020; Online ahead of print.
IVUS: What is New?

David A. Chalyan, MD, MSc
Medical Safety Officer, Chief Medical Office
Director of Clinical Development, Evidence Transformation
Philips Healthcare, Image-Guided Therapy Devices (IGTD)

Denysse Castañeda
Senior Global Product Marketing Manager
IVUS Franchise
Philips Healthcare, Image-Guided Therapy Devices (IGTD)
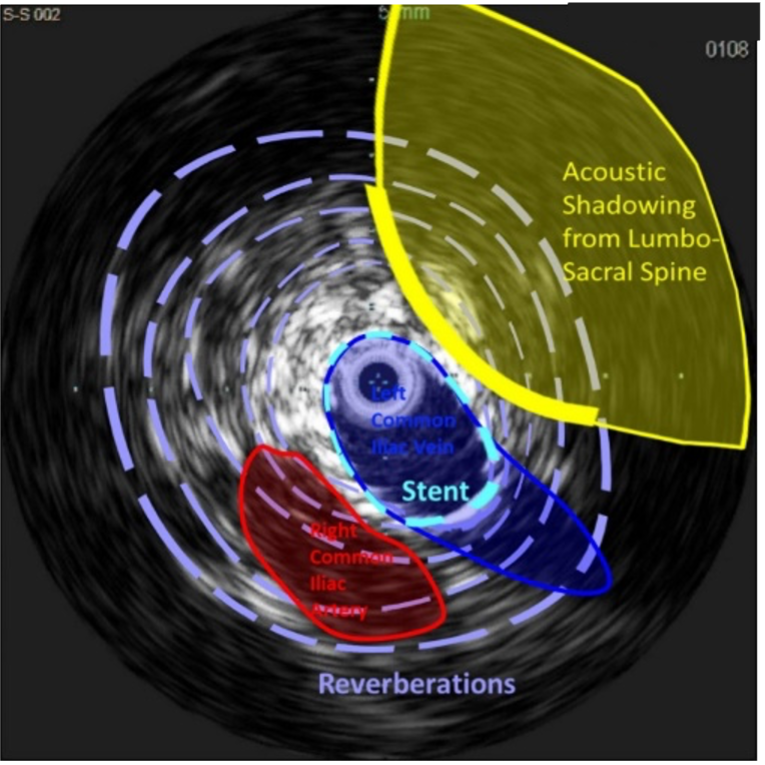
Chronic venous disease (CVD) is a mounting and underrecognized disorder that often presents with symptoms mimicking other medical conditions and can contribute to excess mortality, morbidity, and lower quality of life for patients. Without the proper tools, physicians may not get the insights they need to reliably confirm the diagnoses and plan optimal treatment. The use of Intravascular Ultrasound (IVUS) allows clinicians to visualize disease from within the vessel structure, allowing them to see clearly, assess accurately, and plan precisely. Below are some excellent case-examples of how intravascular ultrasound can improve the procedural outcomes via superior depth and resolution of the vasculature as well as adjacent tissue structures. It is critically important to correctly determine the etiology of compression and accurately size the stent to limit post-procedural complications.
Click images to enlarge.
What is new in the world of venous IVUS? Where do these fit in with the analysis of patients with venous disease?
What we are seeing is a shift in thought on the role and benefits of Intravascular Ultrasound (IVUS) in contemporary vascular procedures. In 2017, the results of the first prospective multicenter study, VIDIO (Venography versus intravascular ultrasound for diagnosing and treating iliofemoral vein obstruction), were published showing the increased sensitivity of IVUS in comparison to multiplanar venography (1). More than half of treatment plans changed after IVUS use with a majority of significant lesions left undetected by multiplanar venography. This shift has also been driven by a number of more recent Real-World Evidence (RWE) studies showing a substantial impact on hard outcomes across vascular beds, including in chronic venous disease patients (2-4).
A large-scale CMS study presented at TCT 2021 by Dr. Eric Secemsky et al found a 31% reduction in the composite health outcome of repeat intervention, hospitalization, or death (4). In addition, it has been remarkable to see such profound associated reductions in risks of stent thrombosis, embolization, and stenosis resulting in improved patient outcomes (4).
The benefits of IVUS in lower extremity iliofemoral intervention is also backed up by the newly released Systematic Literature Review (5) and echoed in the Global Expert Review (6) and Expert Consensus documents presented at VIVA 2021 (7). These studies have supported the systematic utilization of IVUS in practice and led to a number of societal roundtables reviewing the appropriate use and appropriate reimbursement for this technology.
Where do we want the technology to take us in the future?
The high adoption rate of IVUS in iliofemoral venous interventions is driving a robust innovation pipeline in the intravascular imaging space. Philips is committed to new advancements for IVUS technology by finding ways to improve the workflow and making it easier to use. Simplifying the image interpretation of IVUS for new operators will allow for increased use of the technology, quicker diagnosis and better treatment of patients overall.
Our product roadmap is built around the diverse needs of our operators across various specialties. Delivering technology that aids confidence and speed in clinical decision-making, increasing patient access to care through cost-effective, sustainable technology and rapidly innovating technology ecosystems that allow for seamless integration and leading to improved workflow.
References:
- Gagne PG, Tahara RW, Fastabend CP,et al. Venography versus intravascular ultrasound for diagnosing and treating iliofemoral vein obstruction. J Vasc Surg:Venous and Lym Dis 2017;5:678-87.
- Maehara A. Intravascular Imaging to Guide Percutaneous Coronary Intervention Will Be Mandatory Soon. Circ Cardiovasc Interv. 2022 Jun;15(6):e012120. doi: 10.1161/CIRCINTERVENTIONS.122.012120. Epub 2022 May 11. PMID: 35543140.
- Mentias A, Sarrazin MV, Saad M, Panaich S, Kapadia S, Horwitz PA, Girotra S. Long-Term Outcomes of Coronary Stenting With and Without Use of Intravascular Ultrasound. JACC Cardiovasc Interv. 2020 Aug 24;13(16):1880-1890. doi: 10.1016/j.jcin.2020.04.052. PMID: 32819477; PMCID: PMC7444477.
- Secemsky EA, et al. TCT. 2021. (Ahead of Print, Accepted at JACC: Cardiovascular Interventions)
- Natesan S, Mosarla RC, Parikh SA, Rosenfield K, Suomi J, Chalyan D, Jaff M, Secemsky EA. Intravascular ultrasound in peripheral venous and arterial interventions: A contemporary systematic review and grading of the quality of evidence. Vasc Med. 2022 May 12:1358863X221092817. doi: 10.1177/1358863X221092817. Epub ahead of print. PMID: 35546056.
- Secemsky EA, Parikh SA, Kohi M, Lichtenberg M, Meissner M, Varcoe R, Holden A, Jaff M, Chalyan D, Clair D, Hawkins B, Rosenfield K. Intravascular ultrasound guidance for lower extremity arterial and venous interventions. EuroIntervention. 2022 Apr 19:EIJ-D-21-00898. doi: 10.4244/EIJ-D-21-00898. Epub ahead of print. PMID: 35438078.
- Secemsky EA, et al. VIVA. 2021. (Ahead of Print, Accepted at JACC: Cardiovascular Interventions)
CT/MR Venography: When is It Helpful; When is It Maybe Helpful; When is It Unhelpful?

I recently saw a new patient in wound clinic whose presentation, at first glance, seemed to be relatively routine. He was a 79-year-old previously active man with history of left lower extremity deep vein thrombosis (DVT) diagnosed approximately 5 months prior, as well as presenting with current smoking, atrial fibrillation, and chronic heart failure (CHF). His original DVT was likely misdiagnosed as a CHF exacerbation, but ultimately was discovered on ultrasound. He was started on Eliquis® but did not improve. Eventually, he developed a wound related to uncontrolled left lower extremity edema; was treated with compression and antibiotics; and was recommended to present to the wound center.
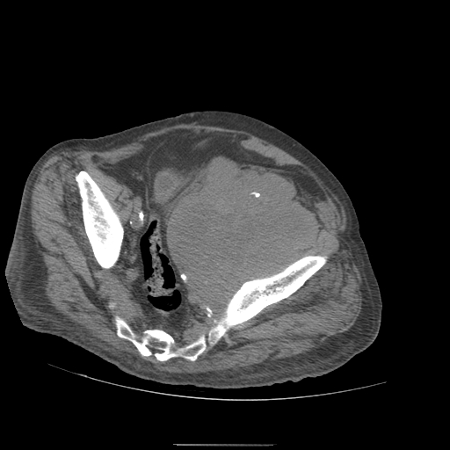
Before his appointment, he presented to the emergency room for worsening lower extremity pain where he was again noted to have (now chronic) left leg DVT. What was unusual about his presentation was that he experienced this unprovoked DVT at the age of 79. Further, he had unusual laboratory findings including a significant acute kidney injury (AKI) thought to be related to poor oral intake and anemia. He finally went to the wound center where he started Unna’s boots and was ordered some further noninvasive arterial and venous testing. However, prior to these tests, he presented to the emergency room again with recurrent falls. By this time, he was experiencing severe fatigue and weight loss. At that presentation, he underwent a CT scan which revealed a large pelvic mass (over 18 cm) encasing his left external iliac and common femoral artery and vein and involving the psoas muscle with bony metastases (Figure 1). He ultimately chose to forgo further work up and entered hospice care.
This unfortunate gentleman’s case represents the challenge of using CT venography and MR venography for the work-up of deep venous disease. While there is likely minimal role for CT/MR venography in the work-up of superficial venous disease (save for possibly some arteriovenous malformations which are beyond the scope of this article), it certainly plays an important role in deep venous evaluation. However, does every patient need it? Probably not. These studies are overused and often misleading as a number of “silent” lesions can be identified on these studies. They play a specific role in the evaluation of deep venous pathology and their appropriate use can save many headaches.
When is CT venography helpful?
- One major benefit of CTV is that it is quick and widely available. However, as many users know, particularly in indirect CTV where contrast is injected into an arm vein, timing of contrast can be suboptimal for detailed venous evaluation.
- Nevertheless, it is excellent for ruling out compressive lesions and finding surprises such as unknown indwelling IVC filters and previously placed stents.
- It can also overcome some of the shortcomings of Doppler ultrasound including the inability to accurately visualize the iliac venous segment due to depth, tortuosity, or acoustic shadowing.
- While CTV has been criticized for inaccuracy compared to the gold-standard IVUS, Raju et al. showed that it has excellent sensitivity, positive predictive value, and accuracy of CTV to detect the presence of venous stenosis. However, the degree of stenosis may be less readily evaluated by this modality.
- In patients with unilateral leg swelling that have a low pre-test probability of May-Thurner syndrome by history and demographics or no other explanation for new lower extremity unilateral swelling, it can be a very useful test for ruling out other possible causes of leg swelling (as in our case above).
When is CT/MR venography possibly helpful?
- When pre-test probability of a May-Thurner lesion is high and when good ultrasound imaging is available, it is unlikely that CTV will add anything but additional radiation to the work-up. In the absence of unusual symptoms, abnormal age of presentation, or other concerning findings on physical exam, proceeding straight to invasive venography with IVUS is likely the best course of action if considering treatment.
- There may be a role for CTV in evaluating the extent of ileofemoral thrombus in the setting of acute DVT. However, this certainly may not be necessary when skilled ultrasonographers are available.
When is CT venography unhelpful?
- CTV can be unhelpful for planning the details of an intervention. Stenotic lesions that may be visualized with IVUS, such as long Rokitansky-type lesions, can be easily missed leading to poor choice in stent landing zones.
- CTV is unhelpful in understanding flow dynamics which are more readily assessed on Doppler ultrasound or invasive venography including pressure measurements, flow direction and phasicity, and direction of flow.
- Moreover, the radiation dose from CTV is not trivial. It should not be used for routine surveillance of stents or known deep venous pathology.
- In the setting of bilateral lower extremity edema with other probably causes, CTV is also unlikely to be beneficial.
- CTV is subject to overuse, especially in the setting of a normal Doppler ultrasound exam. When obtained for patients with atypical symptoms, silent lesions can be discovered which can lead to unnecessary concern and procedures.
Should I use CT or MR venography?
- MRV is subject to the majority of the pros and cons listed above for CTV. It is, however, more expensive and less readily available compared to CTV. It can, nevertheless, provide superior imaging of osteophytes and other bony compressive lesions or in the setting of artifact from orthopedic hardware. It has less radiation, which is an important consideration especially when imaging many of the young female patients that are being evaluated for deep venous pathology.
References:
- Knuttinen, M-Grace, Sailendra Naidu, Rahmi Oklu, Scott Kriegshauser, William Eversman, Lisa Rotellini, & Patricia E. Thorpe. “May-Thurner: diagnosis and endovascular management.” Cardiovascular Diagnosis and Therapy [Online], 7.3 (2017): S159-S164. Web. 22 Jun. 2022
- Jayaraj A, Raju S. Three-dimensional computed tomography venogram enables accurate diagnosis and treatment of patients presenting with symptomatic chronic iliofemoral venous obstruction. J Vasc Surg Venous Lymphat Disord. 2021; 9:73-80.e1. doi: 10.1016/j.jvsv.2020.07.012. Epub 2020 Aug 12. PMID: 32800980.
- Raju S, Walker W, Noel C, Kuykendall R, Jayaraj A. The two-segment caliber method of diagnosing iliac vein stenosis on routine computed tomography with contrast enhancement. J Vasc Surg Venous Lymphat Disord.2020; 8:970-977. doi: 10.1016/j.jvsv.2020.02.021. Epub 2020 May 13. PMID: 32414674.
- Zucker EJ, Ganguli S, Ghoshhajra BB, Gupta R, Prabhakar AM. Imaging of venous compression syndromes. Cardiovasc Diagn Ther. 2016; 6:519-532. doi: 10.21037/cdt.2016.11.19. PMID: 28123973; PMCID: PMC5220205.
- Ghaye B, Szapiro D, Willems V, Dondelinger RF. Pitfalls in CT venography of lower limbs and abdominal veins. AJR Am J Roentgenol. 2002; 178:1465-71. doi: 10.2214/ajr.178.6.1781465. PMID: 12034620.
- McDermott S, Oliveira G, Ergül E, Brazeau N, Wicky S, Oklu R. May-Thurner syndrome: can it be diagnosed by a single MR venography study? Diagn Interv Radiol. 2013; 19:44-8. doi: 10.4261/1305-3825.DIR.5939-12.1. Epub 2012 Jul 16. PMID: 22801870.
- Wolpert LM, Rahmani O, Stein B, Gallagher JJ, Drezner AD. Magnetic resonance venography in the diagnosis and management of May-Thurner syndrome. Vasc Endovascular Surg. 2002; 36:51-7. doi: 10.1177/153857440203600109. PMID: 12704525.
Major Concerns About Venous Imaging in the Routine Clinical Practice

Nicos Labropoulos, PhD
Professor of Surgery and Radiology
Director, Vascular Laboratory
Stony Brook Medicine
A great number of imaging tests are performed every day to evaluate patients with chronic venous disease (CVD). The most common test is ultrasound as it is cheaper, faster, can be portable, and causes no harm. However, several problems exist with the way imaging tests are being performed and interpreted.
Many tests are performed without a complete history and physical exam. Experience is often lacking in performing the tests and miscommunication of what is asked and what is delivered may occur. For example, a CTV is ordered to examine the IVC and iliac veins for obstruction, but only the arterial phase was obtained; or an ultrasound is performed to evaluate reflux in the lower extremity veins and the report focuses on the saphenous veins when the patient presented with non-saphenous varicose veins that extend into the pelvis.
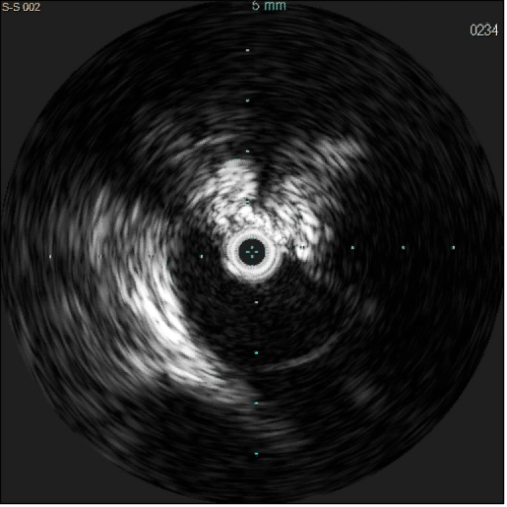
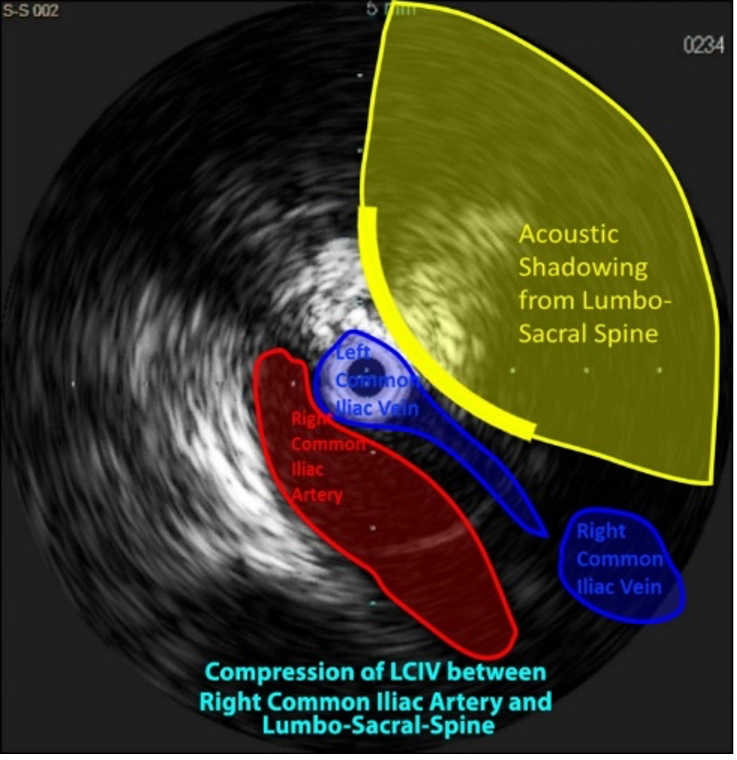
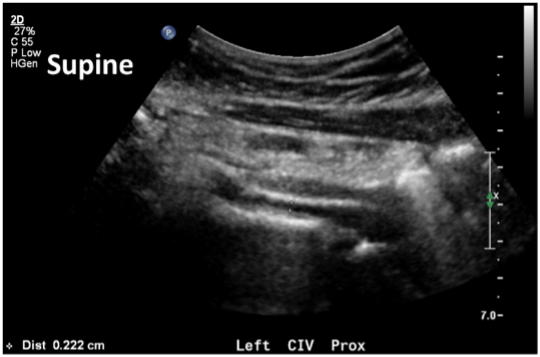
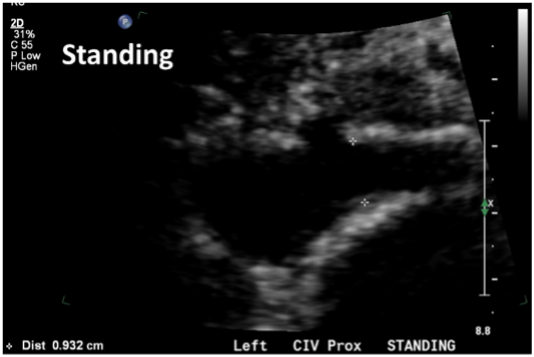
Most imaging tests are performed with the patient in the supine position when evaluating obstruction and reflux in the suprainguinal veins and in many places for detecting reflux in the lower extremity veins. The abdominal, pelvic, and lower extremity veins have their largest diameter during standing. More importantly, CVD symptoms may occur during standing or physical activity; thus, the examination in supine position may not be representative and could also be misleading. Patients with non-thrombotic iliac vein compression may have a tight stenosis in the supine position whereas the stenosis is eliminated on standing (Figure 1). This has become evident in our clinical practice; therefore, we are performing a study to understand the magnitude of this condition. Positional vein stenosis has been shown for both the renal and the iliac veins using IVUS.1 Iliac vein stenosis with collateral formation has been shown using venography in young healthy volunteers.2 Based on these findings, many stents are being placed in patients who have no need for them. Further, many patients with such lesions are treated for having 50% area reductions on IVUS, which is 29% diameter stenosis.
Figure 1. Female patient 28 years old with BMI of 20, and no children, presented with chronic pelvic pain. Symptoms were worse during standing. CTV demonstrated a tight stenosis in the left common iliac vein. She had an ultrasound demonstrating a tight left common iliac vein stenosis in the supine position with a diameter measuring 2.2mm and a vein velocity ratio >4 (left panel). On standing position (right panel) the iliac vein diameter at the same location was 9.3mm without any velocity changes.
Together, with the observations above the low threshold for treatment, they show how serious this issue is.3
Another important issue is using any reflux duration to perform treatment. Our paper on reflux definition in the lower extremity veins is used as an international standard for defining abnormal valve function.4 However, many practitioners use the abnormal value as license to treat. So, many patients with short reflux duration such as 0.7s get treated! Then we can wonder what the impact of a few drops of blood in developing CVD symptoms is! CVD is very prevalent and, as such, many patients will benefit from being treated for the right reason. The role of imaging is paramount for planning and performing the treatment. Rigorous training and experience are necessary to obtain optimal imaging and treatment. Clearly, we need to improve many areas to bring CVD management to the next level.
References:
- Krzanowski M, Partyka L, Drelicharz L, et al.. Posture commonly and considerably modifies stenosis of left common iliac and left renal veins in women diagnosed with pelvic venous disorder. J Vasc Surg Venous Lymphat Disord. 2019; 7:845-52.
- van Vuuren TMAJ, Kurstjens RLM, Wittens CHA, van Laanen JHH, de Graaf R. Illusory angiographic signs of significant iliac vein compression in healthy volunteers. Eur J Vasc Endovasc Surg. 2018; 56:874-9.
- Esposito A, Charisis N, Kantarovsky A, Uhl JF, Labropoulos N. A comprehensive review of the pathophysiology and clinical importance of iliac vein obstruction. Eur J Vasc Endovasc Surg. 2020; 60:118-125.
- Labropoulos N, Tiongson J, Pryor L, et al. Definition of venous reflux in lower-extremity veins. J Vasc Surg.2003; 38:793-8.
The Future of Novel Thermal Imaging in Venous Disease

Ariel Soffer, MD, FACC

Shayna N. Soffer

David Wright, MB, FRCS

Sarah Melvin, MSPH
The use of Duplex Ultrasound (DUS) as a non-invasive diagnostic tool for lower extremity venous disease is the most common and widely preferred method for diagnosing chronic venous insufficiency (CVI) (1,2). However, DUS imaging for CVI is both costly and technically demanding when trying to get an accurate assessment by a highly skilled sonographer and physician interpreter. The DUS exam requires the sonographer to apply manual pressure to visualize flow which causes a need for close contact with the patient for extended, sometimes uncomfortable, periods of time. If the patient cannot stand for the entire exam or the technician cannot physically examine all of the potential areas for CVI, the results of the test may be inaccurate or incomplete (3). Furthermore, current vascular ultrasound training typically concentrates on the arterial and deep venous system with limited time spent in the superficial venous system. These reasons all contribute to why complete superficial CVI mapping is often missed on routine DUS examinations.
Point of Care Thermal Imaging (POCIT) may offer a way to improve these venous mapping inefficiencies. POCIT involves the use of an FDA-cleared infrared camera to detect heat patterns from blood flow near the surface of the skin. The image captured provides a detailed map of the surface temperature, whereby insufficient veins traveling from the deep system are approximately 2-3 degrees Celsius higher than surrounding skin or normal flowing veins. Thermography has been useful in aiding physicians in detecting body surface temperature changes since 1956. POCIT may aid the early detection of certain instances of breast cancer4 and is commonly used in veterinary medicine. More recent research has begun to look at the application of this technology for the diagnosis of chronic venous insufficiency (CVI). Thermal imaging has been found to effectively show a reliable temperature pattern, depicting abnormal venous blood flow, that directly correlates with a clinical finding of CVI and other venous disorders5 shown with DUS.
Figure 1. Venous Insufficiency is about 2 Degrees Celsius greater than surrounding tissue.
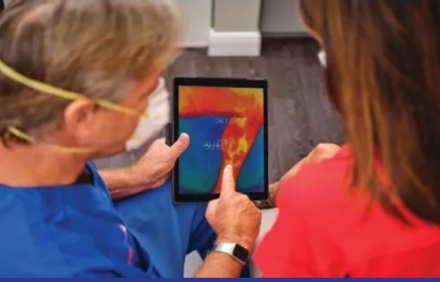
Figure 2. Point of Care Infrared Thermography allows for improved patient understanding.
Adjunctive use of POCIT alongside DUS yields information that is much greater than the sum of the parts alone. This has been shown by multiple small scale studies showing that thermography helps locate CVI not easily seen by DUS. Similarly DUS is able to see CVI that is deeper to the skin, which would not likely be seen by POCIT. However, when both modalities are used adjunctively, optimum imaging and diagnostic power is achieved. Not only can it be used for disease detection, but an easy to understand temperature map often provides an opportunity to close the knowledge gap between patients and physicians. This novel modality may also be considered in the future as a feasible method for efficiently screening patients with suspected superficial venous insufficiency and potentially avoiding the need for ultrasound in the presence of negative thermogram.
References
- Al Shammeri, O., AlHamdan, N., Al-Hothaly, B., Midhet, F., Hussain, M., & Al-Mohaimeed, A. (2014). Chronic Venous Insufficiency: prevalence and effect of compression stockings. International journal of health sciences, 8(3), 231–236. https://doi.org/10.12816/0023975
- Khilnani NM, Grassi CJ, Kundu S, D’Agostino HR, Khan AA, McGraw JK, Miller DL, Millward SF, Osnis RB, Postoak D, Saiter CK, Schwartzberg MS, Swan TL, Vedantham S, Wiechmann BN, Crocetti L, Cardella JF, Min RJ, Cardiovascular Interventional Radiological Society of Europe, American College of Phlebology, and Society of Interventional Radiology Standards of Practice Committees. J Vasc Interv Radiol. 2010 Jan; 21(1):14-31.
- Shabani Varaki, E., Gargiulo, G. D., Penkala, S., & Breen, P. P. (2018). Peripheral vascular disease assessment in the lower limb: a review of current and emerging non-invasive diagnostic methods. Biomedical engineering online, 17(1), 61.
- Lawson R. Implications of surface temperatures in the diagnosis of breast cancer. Can Med Assoc J 1956;75:309-10.
- Bagavathiappan S, Saravanan T, Philip J, Jayakumar T, Raj B, Karunanithi R, Panicker TM, Korath MP, Jagadeesan K. Infrared thermal imaging for detection of peripheral vascular disorders. J Med Phys. 2009 Jan;34(1):43-7. doi: 10.4103/0971-6203.48720. PMID: 20126565; PMCID: PMC2804148.
AVF Abroad & An Update from EVF

Kathleen Ozsvath, MD
Board Member, AVF Board of Directors
Chair, AVF Membership Council
Co-Chair, AVF Diversity, Equity, and Inclusion Committee
The American Venous Forum (AVF) has a long-standing interest in collaborating with international colleagues. Many of these collaborations have grown into mutually beneficial partnerships. The AVF annual meeting each year welcomes members of the European Venous Forum (EVF) in a special session highlighting topics that are of mutual interest to both American and International attendees. Members of the AVF are invited to the EVF in a special session as well. The international committee, chaired by Tomasz Urbanek, is focused on issues specific to the international venous community in general and, more specifically, international AVF members. Areas of interest include research, education, membership, and mentorship. Collaboration to support education for international trainees and venous specialists is of paramount importance. Members of the international committee represent Poland, Mexico, Colombia, Ukraine, England, Italy, Chile, India, and Russia.
I had the distinct honor to attend and participate in the European Venous Forum in Venice, Italy, a few weeks ago. I met colleagues from all over the world who were eager to share their knowledge and expertise. Catching up with Makis Avgerinos, who is working hard in Athens, Greece, and still spending time in Pittsburgh, was great. At the AVF/EVF combined session, I was given the privilege to debate Armando Mansilha in the work up of pelvic venous disease. Armando was eloquent and direct in his points. The following debate was very interesting as well, debating the use of veno-active drugs. Nicos Labropoulos expertly debated with Andrew Nicolaides. I had the pleasure of spending time with friends I have made previously, and many new friends I got to know at the meeting

These opportunities to learn from one another across oceans is imperative to improve the care of patients worldwide. Understanding the difficulties our colleagues face, learning from one another, and creating opportunity internationally makes distance less of an issue and ensures communication and sharing.


Working on International Cooperation Matters

Tomasz Urbanek, MD
Chair, AVF International Committee
Most members of the American Venous Forum are well-aware of the AVF’s significant impact on phlebology development not only in the U.S. but also in many other countries. Widely recognized and implemented guideline documents initiated by the AVF and its members, as well as the research projects, clinical observations, and ideas presented during AVF meetings by AVF members and non-members, confirm an importance of these activities and initiatives not only for the U.S. but also for international phlebology. Let’s take a look at it from the international perspective.
To begin, a short story from my Polish perspective and history: Until the 1990s, there were many excellent physicians in my country, Poland, treating venous diseases, but almost no one used the term “phlebology” in the clinical practice. It should be emphasized that in the end of the 1980s, in Eastern Europe many things were determined not only by medical but also political considerations. In 1988, one of our colleagues, Dr. Tomasz Drażkiewcz, future founder of the Polish Society of Phlebology, worked as a surgeon in Kuwait, where he was successfully infected with the word “phlebology” by Professor Bo Eklöf from Sweden, who was working as a vascular surgeon and phlebologist. Three years later, after contacting other famous European phlebologists, like German Professor W. Hach, Dr. Drążkiewicz founded the Polish Society Phlebology, which is now over 30 years old and progressing in the international field. In the following years, Bo Eklöf (AVF President 2004-2005) visited Poland several times. Now, many years later, our society members take part in AVF annual meetings and other AVF activities.
International members are also an important part of the active AVF members. Currently, 153 international members from 44 different countries actively support the American Venous Forum and represent the international phlebology in the AVF. It is worth mentioning that new members continue to join, which encourages us to make greater efforts to invite phlebologists from around the world to join the AVF community. Among the new AVF members in March and April, we would like to welcome especially our colleague from Ukraine, who joined us during this difficult time for his country.
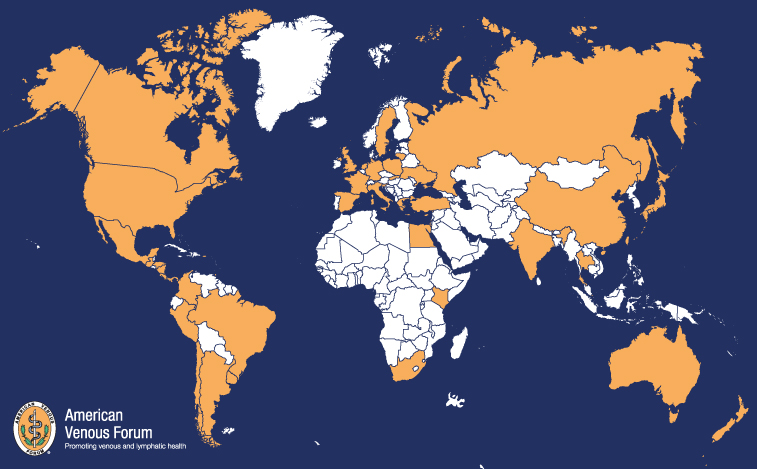


Photo from International Session at VENOUS2022 in Orlando, FL
Both the AVF and the AVF International Committee will continue efforts to increase international collaboration and develop projects which will involve the international community. Among already existing projects such as AVF International Ambassador Program, collaboration with individual international phlebology societies and new projects of international cooperation are currently under evaluation. We look forward to welcoming new AVF international members, and we strongly confirm that we are “working on international cooperation matters!”
Best wishes from AVF International Committee!
American Venous Forum and Latin America United

Alvaro Orrego, MD
Member, AVF International Committee
I feel it is necessary to think about the current relations and the development of future ones in both the scientific and human aspects between the American Venous Forum and the different Latin American scientific societies. What a better way to do this than through a personal experience that can be shared with others.
I remember with nostalgia the first AVF Annual Meeting I attended. I never thought that moment would determine my professional life. I have made a great number of friends who have taken me in with much kindness.
Learning high quality science from great teachers added to the rich practice that we usually carry out in Latin America, building the professional guideline on how we could solve the problems of our patients in a very effective way.
Later came the possibility of becoming a member of the AVF. This was not difficult due to the warm and generous reception from the group, and I quickly felt proud of being part of this institution. There was a feeling of belonging, fraternity, and pride. In some ways, this made me change how I saw our profession.
Why am I making these personal comments? The answer is simple: If I could, anyone can. And what can we do? We can participate in the activities that take place within the institution, and above all, we can contribute with new visions so that together we can make our institution grow in the best possible way. We can generate together new standards, expanding them like boarding an airship to global stages and sharing an already sculpted spirit with patience, humility and effort.
The best growth is the one given by different actors, who in a synergistic work represent the most perfect of the artworks. Without a doubt, during these last years, the AVF has been able to acknowledge the unheard value of excellence of the Latin American professionals. I strongly believe Latin America has been contributing to the AVF with great creativity and excellence.
One of the fundamental AVF activities for us is the Annual Meeting due to its well-recognized quality. The presence of members at the AVF Annual Meeting from other latitudes in our Latin American scientific activities marks a good and honest communication and exchange of experiences through their outstanding speakers.
The scientific endorsement of the AVF in our Latin American meetings held by our accredited scientific societies allows us to give it a seal of seriousness and quality desired by all. Latin America lies in its well-qualified professionals and their inexhaustible desire to work. These professionals can contribute with their diverse experiences, collaboration, and growth of members.
Today, the relationship between Latin America and the AVF is still young, leaving much space to be filled in by further joint work.
“AVF and Latin America working together under the same sky.”
Appropriateness — EVF Congress 2022 in Venice, Italy

Dominik Heim, MD
Past President, European Venous Forum
The European Venous Forum (EVF) Congress in Venice, Italy, was an outstanding meeting held under the EVF President Oscar Maleti and hosted by the Italian College of Phlebology under the President Marzia Lugli.
Though no motto for the congress was given, there was a general feeling that appropriateness is an important debate nowadays. Lots of innovations have been achieved and new technologies have come to market in recent decades, but the paramount question is now their correct use in phlebology patients.

Venous thromboembolism is such a topic: Is it now mechanical thrombectomy (CLOUT Registry)? Is it catheter directed thrombolysis in pulmonary embolism (Sunset trial)? Concerning investigations, which patient with chronic venous insufficiency needs further pelvic imaging? Do patients with chronic venous disease benefit from the use of venoactive drugs? (The two topics discussed in a thrilling pro and contra debate in the joint EVF/ AVF session.) Which patient with iliac outflow obstruction does really benefit from stenting? Do we overstent? This appropriateness was also discussed when comparing the most recent guidelines with that of the ESVS, published at the beginning of 2022. Someone wisely concluded: Think twice before you do it once.

Apart from these animated discussions (one of the cornerstones of each EVF congress is the 10-minute-presentation of the paper followed by 10 minutes of rigorous, merciless discussion), papers that have been submitted for the traditional EVF prize and are evaluated by topic, content, scientific value, and oral presentation. The winner this year was Elisa Munari from Modena with her outstanding paper Hybrid Ilio-femoral Recanalization after Injection for Drug Abuse Obstruction: A Singe Center Experience. Another prize was handed out for the best poster, presented electronically as the usual socializing in front of the posters hung on the wall was not possible. Two honorary members have been elected this year: Sabina Villalta from Italy and Ismail Elalamy from France.
The congress in Venice was – due to lingering Covid – a hybrid congress, with about 450 in-person attendees and around 180 virtual attendees. The challenge to organize a hybrid meeting was met perfectly, demonstrating that IT continues to play an important role in the congresses today, but also confirming its impact on the costs.
EVF continues will continue to host annual meetings, each time in another European country. EVF 2023 will take place in Berlin, Germany, from June 22-24, 2023, under the auspices of the German Phlebological Society with the congress President Erika Mendoza. With interest, we look forward to more of these challenging papers and, of course, another animated EVF/AVF debate, that shows that we sometimes (e.g. venoactive drugs) deal differently with the same subject.
Let’s continue to look for the appropriate treatment for our phlebological patients on both sides of the Atlantic.
Though no motto for the congress was given, there was a general feeling that appropriateness is an important debate nowadays. Lots of innovations have been achieved and new technologies have come to market in recent decades, but the paramount question is now their correct use in phlebology patients.