Table of Contents
Message from the Editor: Pontch, Rosie, and Zebras
Steve Elias, MD, Editor-in-Chief
Membership Matters
Membership Drive 2022: Recruit a New AVF Member Today!
Mona Li, MD, Chair of AVF Membership Committee
Venous For All: Together in DEI
Karem Harth, MD, MHS, RPVI and Anil Hingaroni, MD
Odds & Ends of Venous Disease
Peripheral Venous Aneurysms
Thomas W. Wakefield, MD, and Peter F. Lawrence, MD
Portal Vein Aneurysms
Mary Binko, MS, and Eric Hager, MD
IVC Cross-section and Hydration
David M. Williams, MD, and Andrea Obi, MD
The Iceberg Phenomenon that is Klippel-Trénaunay Syndrome (KTS)
Michael Dalsing, MD, MBA, DFAVF
Common Dermatologic Diseases that Masquerade as Venous Pathology
Margaret Mann, MD, FAAD
Pediatric Thrombosis: Not Just Miniature Adults!
Joann Lohr, MD, FACS, RVT, CWSP
The Underreported Morbidity of Mesenteric Venous Thrombosis
Elizabeth Andraska, MD, MSc, and Natalie Sridharan, MD
When is DVT not Just a DVT?
Chandu Vemuri, MD and Robert Beaulieu, MD, MSE
Publications and Hoofbeats
Ruth L. Bush, MD, JD, MPH
International Corner
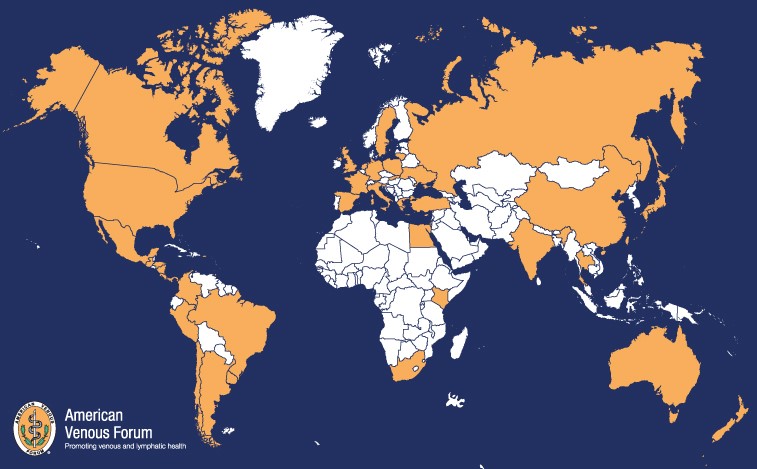
AVF Representation Across the Globe
Swiss Society of Phlebology Highlights: Diversity in Phlebology
Jürg Hafner, Dominik Heim, Jürg Traber
Copyright © 2022 by The American Venous Forum. All rights reserved.
EDITOR-IN-CHIEF
Steve Elias, MD
EXECUTIVE EDITOR
John Forbes, MBA
MANAGING EDITOR
Allie Woodward
PUBLICATION DESIGNER
Christine Rataj
Message from the Editor: Pontch, Rosie, and Zebras

Steve Elias, MD
Editor-in-Chief, Vein Specialist
Cat ice cream is probably next. As far as I’m concerned, this is a pure marketing ploy. My wife and I have had many dogs throughout our marriage. We gave them a spoonful of human ice cream when we were eating some. They liked it. They didn’t have GI upset. Ben & Jerry think that this is a real doggie problem, GI upset.
Do dogs really need ice cream–human or doggie? If you never give them ice cream, they will never know what they’re missing. My experience has been that dogs will eat almost anything; well maybe not asparagus, mushrooms, or peas. But a lot of humans won’t eat them either. Do we really need Ben & Jerry’s Doggie Dessert ice cream in 2022? There are some humans with elevated BMIs who don’t really need it either. It is not an essential nutrition staple. It is a treat. It’s all about perspective.
Go to the Ben & Jerry’s website. They almost convinced me that my Corgi needed their sunflower butter based doggie ice cream. Breaking news… they say humans can eat it, too. Wow. What will your friends think as you lick either Pontch’s Mix or Rosie’s Batch doggie ice cream? Pontch and Rosie are Ben & Jerry’s dogs. When Pontch and Rosie create human ice cream maybe they’ll name it Ben’s Dream and Jerry’s Joy. Ludicrous.
In this issue of Vein Specialist we decided to go a little off the beaten path, (Clearly, Ben & Jerry are way off.) We have dedicated this issue to venous zebras: the unusual, uncommon pathologies that are the odds and ends of our specialty. Some of us rarely see them and some of us may never see them. We have 2 articles about aneurysms – portal and peripheral. DVT in unusual places or people is discussed by Chandu Vemuri and Joann Lohr – mesenteric veins and pediatric population. Margaret Mann, our resident dermatology person, talks about some dermatologic conditions that can mimic venous skin findings. Michael Dalsing’s article discusses that the skin components of KTS are merely the tip of the iceberg. Finally, our last venous zebra is David Williams and Andrea Obi’s article about IVC morphology/problems and hydration.


Now that we have brought your attention to venous zebras, ever wonder what zebras eat? We know that dogs crave ice cream according to the marketing people at Ben & Jerry’s. They didn’t make ice cream for zebras because zebras aren’t picky eaters. In fact, they’ll eat almost anything green: grass, leaves, asparagus, peas, and perhaps money. Maybe Ben & Jerry just need to create a green ice cream for our Vein Specialist zebras? Until this issue I didn’t much think about zebras. I have a few other things that I need to think about: wife, four children, grandchildren, a job, and–most important–what Doggie Dessert type of ice cream I should bring home for our dog. And I thought decision making when treating patients with venous disease was tough at times! Ben & Jerry take it to a new level. This issue exposed you to some zebras in our vein world. And even though many of us don’t encounter zebras over the course of our professional lives, we hope this issue will help you when you do. Regarding your dogs, just give them a teaspoon of real human ice cream and call it a day. Ben & Jerry’s be damned.
Membership Drive 2022: Recruit a New AVF Member Today!

Mona Li, MD
Chair, AVF Membership Committee
As the AVF embarks on a new membership campaign, I began to reflect on how I became a member of the American Venous Forum.
As a general surgeon, I had the opportunity to begin my career in venous disease with the Division of Vascular Surgery at the University of Rochester in Rochester, NY in 2006. At that time, this division already operated a free-standing vein clinic. The person I credit as my main mentor, Dr Jeffrey Rhodes, encouraged me to train with them to learn the latest minimally invasive techniques for superficial venous disease. Dr Rhodes introduced me to the American Venous Forum, and I have been a member since 2007.
In 2011, when I was on faculty at the University of Cincinnati, Dr.George “Mickey” Meier directed me to the AVF Fellows and Early Career Course to further enhance my venous disease education and ultrasound skills. I followed their advice to become a member and to take advantage of all the benefits that this society provides for those who have an interest in venous and lymphatic disease.
While I can certainly espouse the enormous list of educational, career, and practice-enhancing perks of membership, the camaraderie, friendships, and new mentors that I have met through membership in the AVF have been the most gratifying benefits. Being surrounded by like-minded practitioners and researchers from around the world who share the same vision and purpose in advancing the care of patients with venous and lymphatic disease and increasing awareness of the clinical importance of our field is truly invaluable and worthwhile aspect of membership in this remarkable society.
As Chair of the Membership Committee this year, I want you to reflect on why you are a member of the American Venous Forum, and how you can pass on all the same benefits and rewarding relationships you have received from the AVF to a fellow physician who shares the same commitment to venous and lymphatic disease.
AVF member benefits include but are not limited to:
-
-
- Free online access to Journal of Vascular Surgery: Venous and Lymphatic Disorders
- Reduced registration fees to AVF’s Annual Meeting and other educational programs
- Reduced costs of selected educational publications in AVF’s online store ShopAVF
- Free access to Venous Online® for recorded content and educational programs
- Access to AVF Exchange, AVF’s private discussion board
- Inclusion in VitaDox, an online resource for patients to find a vein specialist
- Access to AVF’s Career Center to learn about new opportunities as a vein specialist
-
If you recruit a new physician member during the months of October and November 2022, you will receive a special bonus for your efforts. Be a mentor and recruit a fellow physician to join the AVF today! The Membership Committee and I thank you for your time and consideration.
Venous For All: Together in DEI

Karem Harth, MD, MHS, RPVI
Member, AVF Early Career Committee
Member, AVF DEI Committee

Anil Hingaroni, MD
Member, AVF Newsletter Committee
Member, AVF DEI Committee
The AVF core values include those of Diversity, Equity, and Inclusion. The commitment to understanding, accepting, respecting, and encouraging all ideas, people, and actions will advance AVF priorities and goals and elevate all aspects of society. Our newly developed ad hoc Diversity, Equity, and Inclusion (DEI) Committee will actively implement and create actionable goals in line with DEI principles. Research that is sensitive to DEI matters is of paramount importance to better understand all populations in an unbiased way. Internal reflection, we believe, will be a catalyst to the process of making meaningful strides in the DEI space. Looking at these data represents an important shift in our priorities and will elevate our society and its mission. To this end, we share voluntary survey results from our membership and report the make-up of our current committees and leadership.
AVF Membership
Our commitment and efforts in the DEI space, importantly, start with our membership as reflected in many areas including representation and role modeling by our AVF leaders, interest in venous disease by physicians and trainees, and how effective we are in demonstrating our inclusivity. These are just some of the ways our membership reflects on us and our actions. In 2021, the AVF performed a voluntary survey of our membership. The self-reported results provide a view of our membership, areas of opportunity, and the makeup of the global venous community as it exists and that we can build upon.

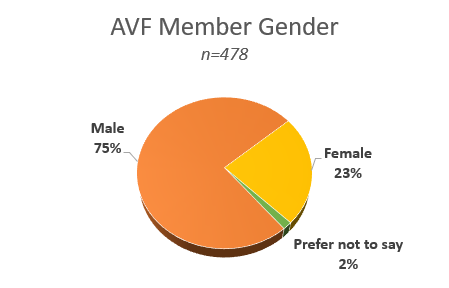
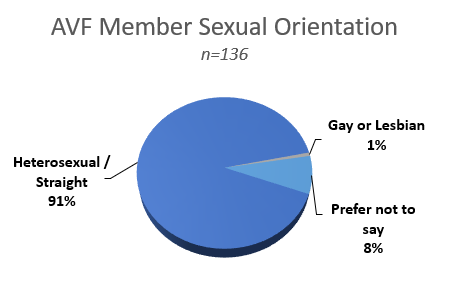
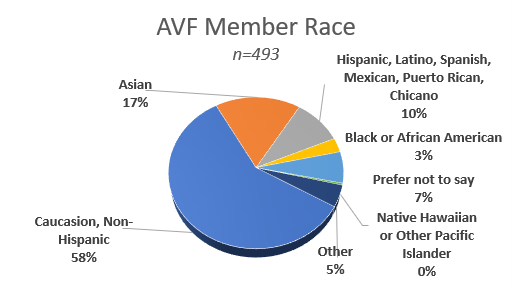
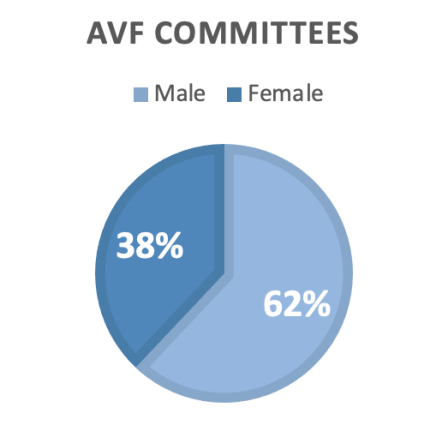
AVF Committees
AVF committees play critical roles in the success, growth, and representation of our society. AVF supports 22 active committees with 194 volunteer positions filled by 118 active AVF members. Across all committees, 62% of the members are men and 38% are women. Of the active AVF members, 57% are men and 43% are women.
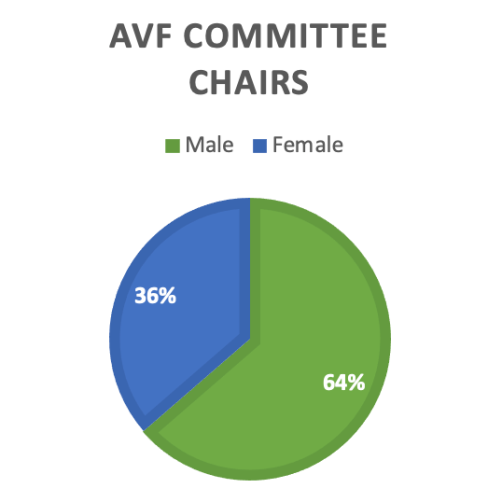
Each of these committees is chaired by a selected AVF member. Chairs of these 22 committees are 64% men and 36% women. In committees chaired by men, 28% of the committee members were women; while in committees chaired by women, 54% of the members were women. Among the 22 committees, 9 committees have equal numbers of men and women members.
Gender balance in specific committees was noted as a factor that will have an impact in the current and future image of the AVF. These committees in particular include mission advancement, digital media, annual program, patient education, membership, research, early career, DEI, and document oversight. We want to encourage engagement and participation and thus we highlight areas where we can be purposeful in our elections and sponsorships. From our 22 committees, those that have been identified as having an opportunity to improve gender-based diversity include international committee, ethics committee, bylaws committee, strategic planning committee, distinguished fellows (selection) committee, nominating committee, finance committee, and executive committee.
The CORE VALUES VEINS emphasizes Diversity, Equity, and Inclusion. As we reflect on these initial data, we want to thank those who voluntarily contributed to this survey as well as our leadership for openly sharing the data with our group and readers. Our membership, committee, and leadership survey provides an important “first look” at our make-up. Future updates to these data will allow us to monitor progress and serve as a surrogate for meaningful impact and change in our DEI efforts. Importantly, they highlight limitations and knowledge gaps in our data that we need to prioritize as we move forward. Our voices will be better heard and represented when we can improve on other important demographic variables not addressed by our current surveys. We need to better understand presence of under-represented minorities, various ethnicities, and LGBTQ groups, to name a few. Promoting equity and diversity starts with Inclusivity, which requires us to cultivate a culture and climate of non-judgment and open conversation. We will build momentum through the process of identifying our strengths and limitations. By increasing awareness, we will cultivate an ideal DEI platform – Venous for All.
Peripheral Venous Aneurysms

Thomas W. Wakefield, MD
Past AVF President
University of Michigan

Peter F. Lawrence, MD
Past AVF President
UCLA Medical Center
Venous aneurysms increase the risk for venous thromboembolism (VTE) and pulmonary embolism (PE) and can lead to the development of chronic venous insufficiency (CVI). A venous aneurysm is defined as a vein that measures 1.5-2.0 times the size of the normal vein below or above the aneurysm and can be either fusiform or saccular. The most common locations for peripheral venous aneurysms are popliteal, iliofemoral, and upper extremity/jugular. In a single center study, 3 (~30%) patients presented with DVT or PE; 3 (~30%) had limb pain or swelling; and 40% were asymptomatic (1). The mean size of popliteal venous aneurysms was 2.6 times the normal proximal and/or distal vein. Of asymptomatic patients who were observed, 3 ultimately underwent surgical repair due to size or symptoms.
In a multicenter study of venous aneurysms from the low frequency disease consortium with a median follow-up of 27 months, 8 patients with popliteal venous aneurysms (20%) presented with DVT/PE; 13 (33%) presented with pain; and the remainder were discovered incidentally (2). Patients presenting with DVT or PE had larger aneurysms than those presenting without VTE (3.8cm vs 2.5cm, p=0.003). Additionally, aneurysm morphology (saccular versus fusiform) was associated with thromboembolism (30% vs 9%, p=0.046) as was thrombus burden, which was determined by duplex ultrasound and/or cross-sectional imaging, with an estimate of intravascular thrombus measuring > 25% (45% vs 3%) being associated with DVT or PE. Because large lower extremity venous aneurysms and saccular aneurysms with thrombus >25% lumen involvement were more likely to present with thromboembolic complications, they deserve operative intervention.
Half of all patients treated for popliteal and iliofemoral venous aneurysms combined underwent immediate intervention, whereas the remainder of patients were managed with observation and an antithrombotic regimen. In the medically managed cohort, the incidence of DVT was 5% for popliteal venous aneurysms, but 33% for observed iliofemoral venous aneurysms. For upper extremity aneurysms, only 1 medically treated patient developed pain and 1 demonstrated growth. Thirty-nine patients (59%) ultimately underwent surgical intervention, with the most common indication for surgery being absolute size. Nine patients underwent a procedure for DVT/PE and 11 for pain or extremity swelling. The most common operation was aneurysmorrhaphy in 21 patients (54%), followed by excision and ligation in 14 patients (36%), and interposition bypass grafting in 5 patients (13%).Postoperative hematoma in anticoagulated patients requiring re-intervention was the most common complication, present in 3 popliteal vein repairs and 1 iliofemoral vein repair. Since postoperative hematoma is the most common complication, the technique of closed plication is often preferred to open plication for popliteal venous aneurysms (and iliofemoral venous aneurysms) if no thrombus is present in the aneurysm, as anticoagulation is unnecessary from the viewpoint of the surgery (3).
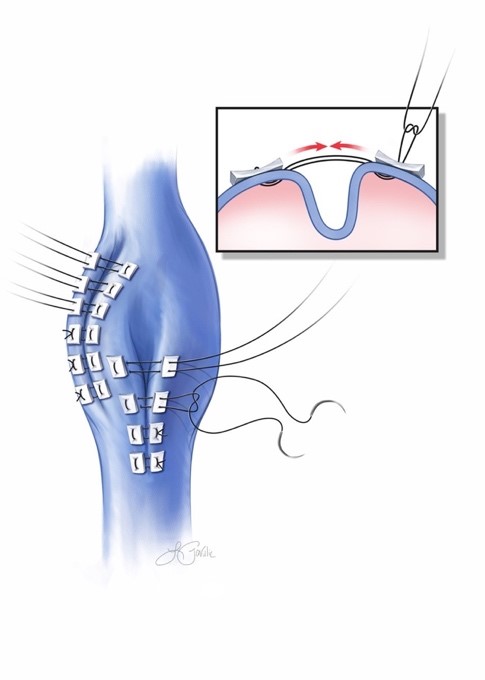
In conclusion, surgical intervention for lower extremity venous aneurysms is indicated to reduce the risk of VTE and need for continued anticoagulation, with popliteal venous aneurysms > 2.5cm and all iliofemoral aneurysms considered for repair. Plication (Figure 1) is the preferred procedure when there is no intraluminal thrombus because postoperative anticoagulation is not required. Upper extremity aneurysms do not carry a significant risk of VTE and warrant treatment primarily for symptoms.
Figure 1. Plication of popliteal vein aneurysm, in this case with two rows of plication sutures. Modified from Beaulieu et al.3 Figure by RJ Beaulieu. Used with permission.
References
1. Patel R, Hanish S, Baril D, Woo K, Lawrence P. Contemporary management of lower extremity venous aneurysms. J Vasc Surg Venous Lymphat Disord. 2019;7:860-864. doi:10.1016/j.jvsv.2019.06.017
2. Patel R WK, Woo K, Wakefield TWW, et al. Contemporary management and outcomes of peripheral venous aneurysms – a multi-institutional study. J Vasc Surg Venous Lymphat Disord. 2022 Aug 5:S2213-333X(22)00326-2. doi: 10.1016/j.jvsv.2022.06.011. Epub ahead of print. PMID: 35940449
3. Beaulieu RJ, Boniakowski AM, Coleman DM, Vemuri C, Obi AT, Wakefield TW. Closed plication is a safe and effective method for treating popliteal vein aneurysm. J Vasc Surg Venous Lymphat Disord. 2021; 9:187-92. doi:10.1016/j.jvsv.2020.04.026
Portal Vein Aneurysms

Mary Binko, MS
University of Pittsburgh

Eric Hager, MD
University of Pittsburgh
Chair, AVF Digital Media Committee
Member, AVF Newsletter Committee
From time to time we receive referrals for portal vein aneurysms, but we do not have consensus recommendations to guide our management of these patients. Due to the rarity of venous aneurysms, our current understanding is based on case reports and series as well as systematic reviews. However, portal vein aneurysms are increasingly reported as an incidental finding on imaging, thus urging us to develop a better understanding of this venous aneurysm.
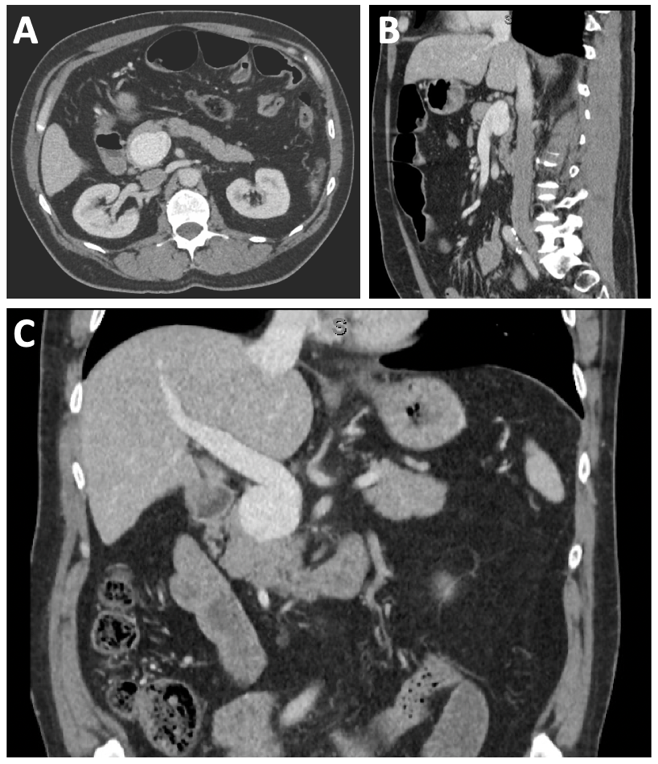
Portal vein aneurysms occur at any age without male or female predominance. Venous aneurysms form due to degeneration and weakening of the vein wall resulting in aneurysmal dilation diagnosed on ultrasound or computerized tomography (Figure 1). This can lead to nonlaminar blood flow resulting in thrombosis or increased wall stress and spontaneous rupture.
The etiology of portal vein aneurysms is acquired or congenital. The most common acquired etiology is liver disease (26-28.3%) and portal hypertension (30.8-32%) (1). However, many patients without liver disease develop portal vein aneurysms. Case reports have described the presence of portal vein aneurysm in the fetus, which supports an underlying congenital origin. Developmental abnormalities in the vitelline and umbilical veins, which form the portal venous system, lead to portal vein aneurysms (2).
No consensus guidelines have been published on the management of portal vein aneurysms. The natural history, including their shape and rate of growth, is poorly understood. In an analysis of 53 patients, 94% of aneurysms remained stable without interval growth over a mean of 21.15 months (3). Consequently, patients with small aneurysms without interval growth on serial imaging are managed conservatively. Several indications for surgical intervention include size, rapid interval growth, symptoms, and complications. The diameter at which intervention is indicated remains unclear due to our incomplete understanding of the natural history of these aneurysms and the size at which risk of rupture outweighs the surgical risks. A size of 3 cm has been suggested as an indication for intervention (4). The most common symptoms leading to intervention are abdominal pain (44.7%) and GI bleeding (7.3%).3 Complications are rare, including thrombosis, rupture, or compression of nearby structures, including the common bile duct, duodenum, and inferior vena cava (1,3).
The presence of portal hypertension influences the selection of operative technique as patients with portal hypertension receive portosystemic shunts for venous decompression, splenectomy, or liver transplant (1). Aneurysmorrhaphy or aneurysmectomy is the preferred operative technique for patients without liver disease and thrombectomy when thrombosis is present. Although open surgical intervention remains the standard of care, a role for endovascular intervention in the treatment of portal vein aneurysm may exist. A hybrid approach using laparotomy and catheter-directed thrombectomy was utilized recently for a case of aneurysm thrombosis (5).
Figure 1. A.) 4.3 x 3 cm fusiform portal vein aneurysm in an asymptomatic patient in A, axial; B.) sagittal; and C.) coronal orientation.
References
1. Laurenzi A, et al. Portal vein aneurysm: what to know. Dig Liver Dis.2015; 47: 918-23. doi:10.1016/j.dld.2015.06.003
2. Gallego C, et al. Congenital and acquired anomalies of the portal venous system. Radiographics. 2002; 22:141-59.doi:10.1148/radiographics.22.1.g02ja08141
3. Sfyroeras GS, et al. Visceral venous aneurysms: clinical presentation, natural history and their management: a systematic review. Eur J Vasc Endovasc Surg. 2009; 38:498-505. doi:10.1016/j.ejvs.2009.05.016
4. Moreno JA, et al. Extrahepatic portal vein aneurysm. J Vasc Surg. 2011; 54:225-6,.doi:10.1016/j.jvs.2010.05.113
5. Gorolay V, et al. Asymptomatic thrombosis of extrahepatic portal vein aneurysm necessitating hybrid operative repair. Vascular. 2021; 29:762-66. doi:10.1177/1708538120976977
IVC Cross-section and Hydration

David’s M. Williams, MD
University of Michigan

Andrea Obi, MD
Chair, AVF Research Committee
Member and Past Chair, AVF Digital Media Committee
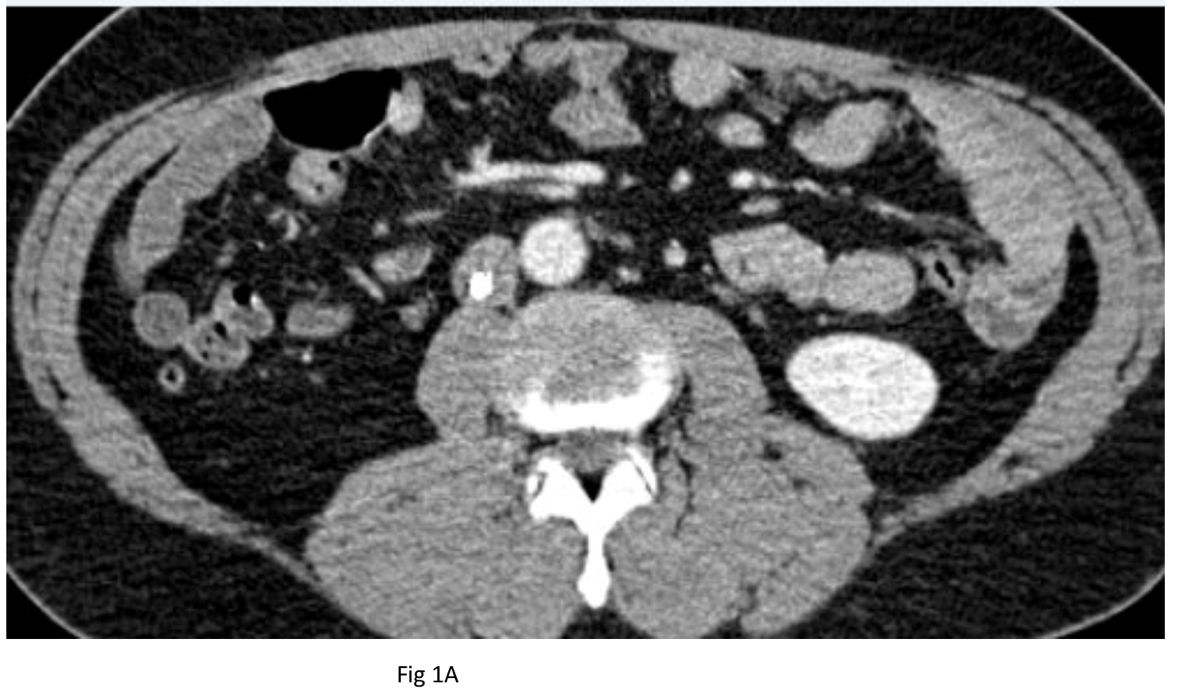
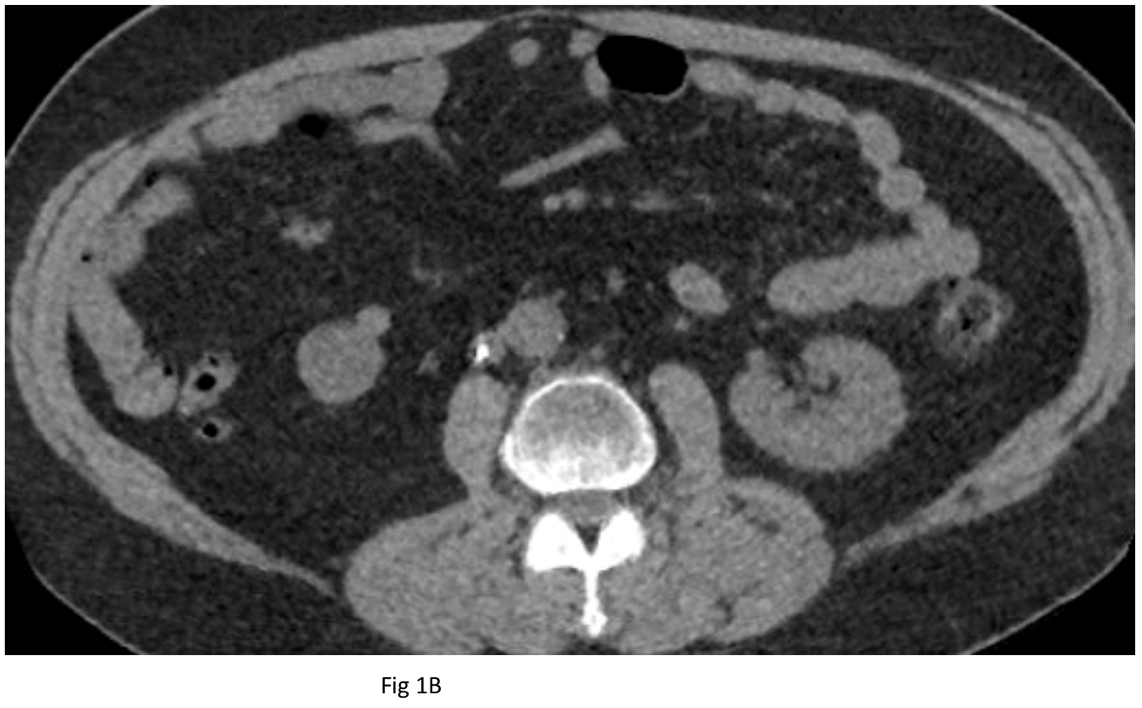
The patient was a 56-year-old man referred to us for removal of a Guenther-Tulip IVC filter placed because of several thromboembolic episodes 10 years earlier. He was found to have the G20210A mutation. Anticoagulation was discontinued after 1 year, and the patient on aspirin only had no recurrence over the intervening years. He had no varicose veins in his legs or abdomen. He had no limitation to his activities of daily living. He walked 30 minutes 3-5 days weekly with no discomfort or limb swelling. After an attempt to remove the IVC filter failed, a CT was obtained, and the patient was referred for filter removal and possible IVC reconstruction.
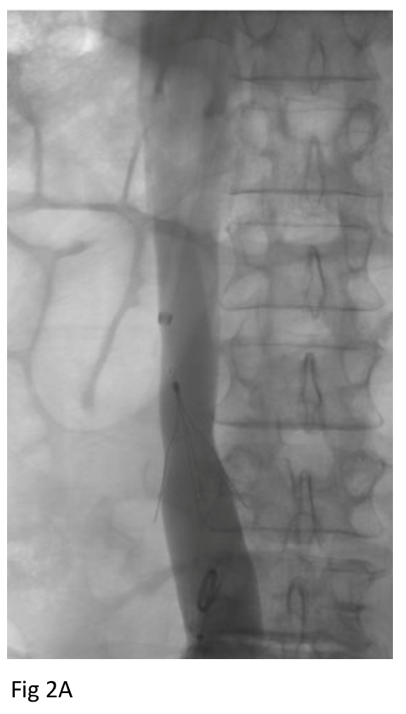
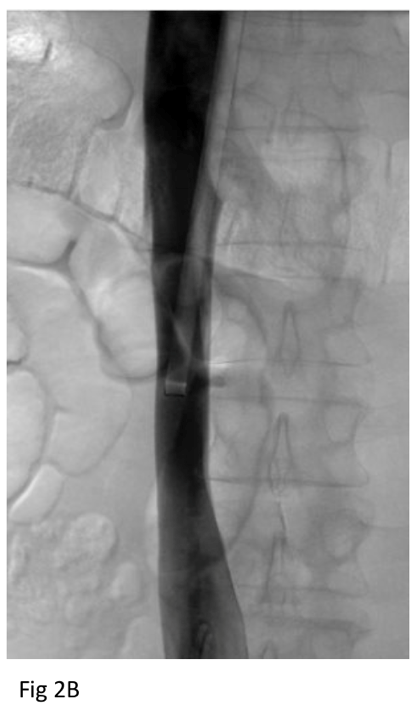
Baseline CT shortly after filter placement showed normal caliber IVC at the level of the filter apex (Figure 1A). CT after failed retrieval 8 years later showed marked narrowing of the IVC at the level of the filter apex (Figure 1B). On the day of filter extraction 3 months later, the patient’s creatinine was 1.6 (baseline 1.3), so he was given 2 liters of lactated Ringer’s. Right trans saphenous iliocavogram after the crystalloid infusion (Figure 2A) showed normal caliber IVC and coaxial filter with no stricture or collateral flow. The IVC filter apex was free in the IVC lumen, and the filter was removed uneventfully using 12 Fr endobronchial forceps through a 16 Fr sheath. Post-extraction cavogram showed excellent flow with no stricture or contrast extravasation (Figure 2B).
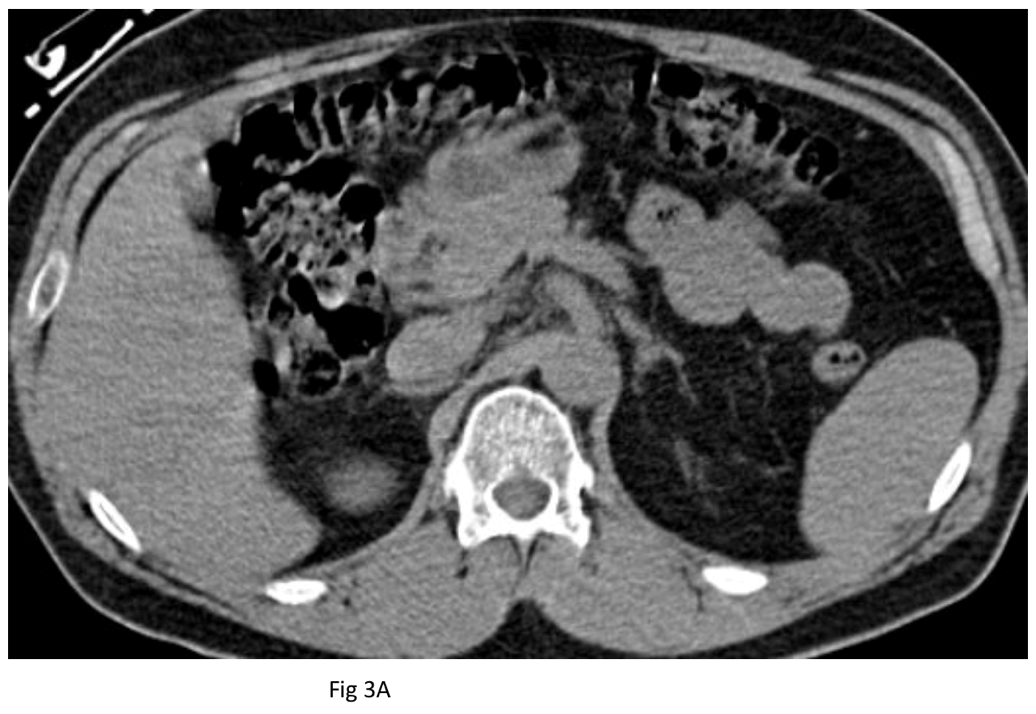
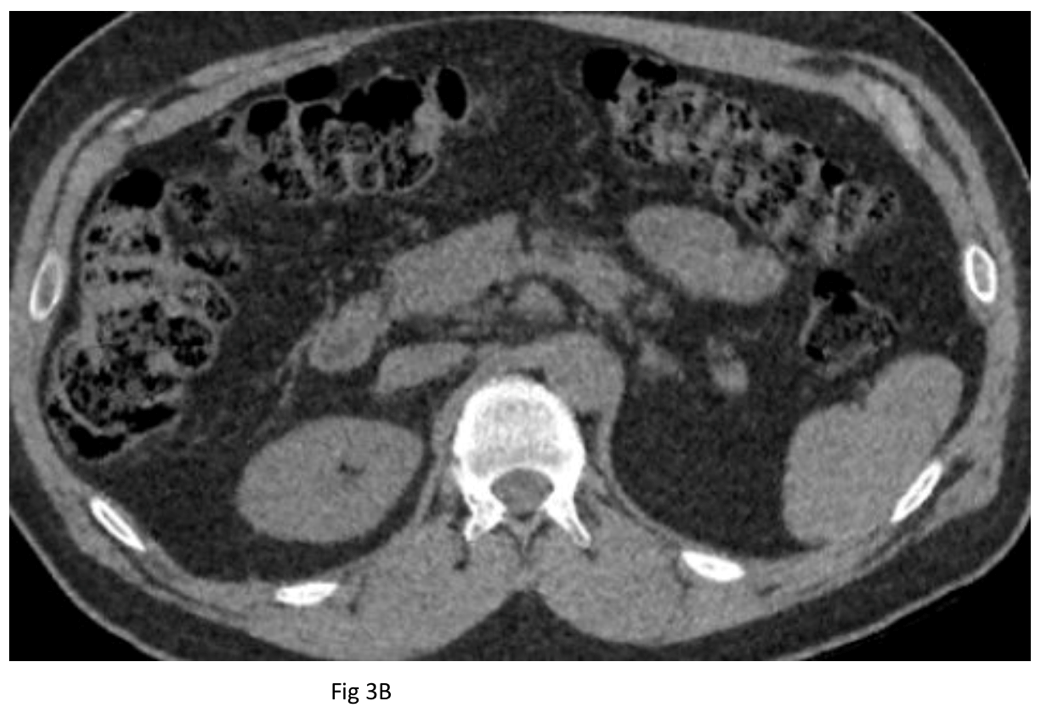
This case illustrates the remarkable change in IVC caliber due to fluid status of the patient. Although we do not have biochemical markers of the patient’s fluid status at the time of exams, the CTs show small caliber suprarenal IVC at the time of failed extraction (Fig 3A) compared to normal caliber IVC after crystalloid infusion (Fig 3B) just prior to successful extraction. Unfortunately, we do not have a creatinine level at the time of the baseline CT, but we presume the change in IVC caliber was due entirely to the added 2 liters of crystalloid he received just before the venogram. Although hydration’s role is not rare in these cases, the factitious role hydration status can play in imaging is often not appreciated yet deserves attention and elaboration.4
Figure 1. CT shows normal IVC and filter apex several days after implantation (A) and contracted IVC several days after initial failed extraction (B) 8 years later.
Figure 2. Cavogram after crystalloid infusion before (A) and (B) after filter retrieval show normal caliber IVC without stricture or contrast extravasation.
Figure 3. From the same exams as in Figure 1 above, axial CT slices show normal caliber suprarenal IVC several days after implantation (A) and contracted IVC several days after initial failed extraction (B).
References
1. Raju, S. Ten lessons learned in iliac venous stenting. Endovasc Today. 2016;15:40-44.
2. Albani S, Mesin L, Roatta S, et al. Inferior vena cava edge tracking echocardiography: a promising tool with applications in multiple clinical settings. Diagnostics (Basel). 2022; 12:427.
3. De Vecchis R, Baldi C. Inferior vena cava and hemodynamic congestion. Res Cardiovasc Med. 2015; 4:e28913. doi: 10.5812/cardiovascmed.28913v2. PMID: 26436075; PMCID: PMC4588705
4. Eisenstat RS, Whitford AC, Lane MJ, Katz DS. The “flat cava” sign revisited: what is its significance in patients without trauma? Am J Roentgen. 2002; 178:21-25.
The Iceberg Phenomenon that is Klippel-Trénaunay Syndrome (KTS)

Michael Dalsing, MD, MBA, DFAVF
AVF Past President
Professor Emeritus, Division of Vascular Surgery
Indiana University School of Medicine
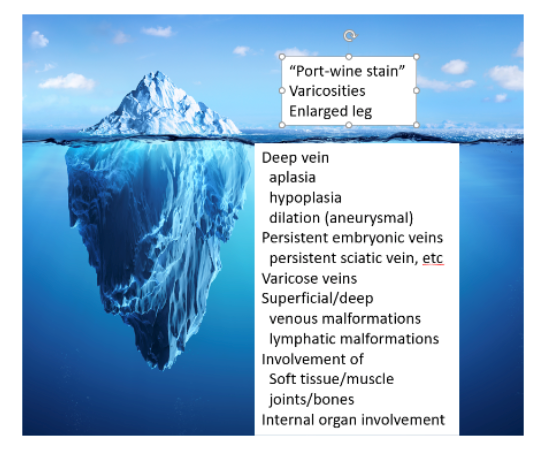
Klippel-Trénaunay Syndrome (KTS) is classified (Hamburg classification) as a predominantly venous defect of the truncular form (signifying the inability to proliferate after removal) presenting with aplasia, obstruction, or dilation.
A venous duplex study provides a non-invasive baseline of the lower extremity venous system. We start to see a glimpse of the more expansive underwater iceberg in persistent embryonic veins (often located laterally, large, and termed the lateral marginal or persistent sciatic vein), superficial venous malformations, varicose veins, deep venous hypoplasia, segmental aplasia, and aneurysmal degeneration. In addition to occlusive disease, venous reflux is often demonstrated.
Magnetic resonance imaging (MRI) further delineates these venous abnormalities and malformations, but also reveals lymphatic malformations not previously suspected and allows an assessment of the involvement of muscle, joint, and bone. It can discriminate venous from the more insidious and life-threatening arterial malformations involved with different diagnoses such as the Parks Weber Syndrome. Involvement of the bones and joints will suggest a need to collaborate with our orthopedic colleagues.
Most patients experience a tolerable disease course managed with compression stockings, manual/pneumatic compression, and moderate lifestyle changes. One consequence of the hidden iceberg effect is the potential for a longer leg that needs the expertise of a prosthetist (foot inserts/special shoes) or, in the young patient, an orthopedic surgeon to correct the difference in leg growth. The abnormal varicose vein(s) can be treated using percutaneous access to allow placement of long catheters or a needle to permanently close the vein with heat, glue, or corrosive agents; but, sometimes, surgery works best. However, care must be taken to make sure that deep vein obstructions are not so extensive that blood cannot get out of the leg after the superficial veins are closed. For the abnormal deep veins that are causing muscle, joint, or bone enlargement or damage, sticking a vein and using small wires and catheters to get to the problem area can allow corrosive agents, glues, or plugs to destroy the abnormal venous collections. A vein specialist can use many tools to expertly understand the iceberg that is the KTS and plan a personalized treatment.
Reference
Wang SK, Drucker NA, Gupta AK, Marshalleck FE, Dalsing MC. Diagnosis and treatment of the venous malformations of Klippel-Trénaunay syndrome. J Vasc Surg Venous Lymphat Disord. 2017; 5:587-95.
Common Dermatologic Diseases that Masquerade as Venous Pathology

Margaret Mann, MD, FAAD
Dermatologic Surgery Specialist
Skin changes are often the first clue to the diagnosis of venous insufficiency. As venous specialists, we are all familiar with the classic dermatologic manifestations of chronic venous insufficiency such as stasis dermatitis, edema, corona phlebectasia, lipodermatosclerosis, and venous ulcers. But not all rashes that present on the lower extremities are venous in origin. Here we highlight 4 dermatologic diseases that masquerade as venous pathology.
Case 1
This 43-year-old male reports this episodic red, minimally pruritic rash along the shins and ankles after prolonged standing. The rash lasts a few weeks then fades away.
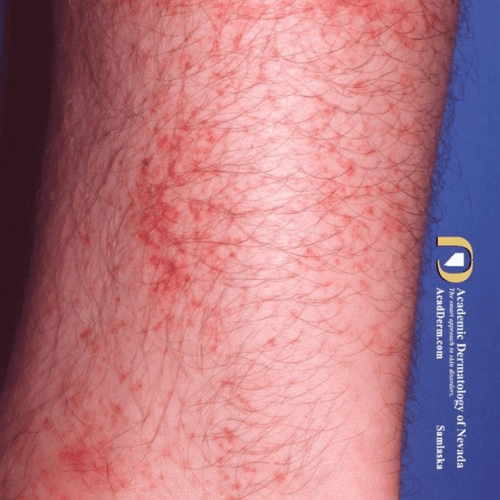
Diagnosis: Schamberg purpura
Capillaritis, or Schamberg purpura, is due to the red blood cell extravasation from tiny capillaries resulting in hemosiderin deposits and the characteristic non-blanching, non-palpable “cayenne pepper” petechiae and rust-colored pigmentation. The cause of the capillary inflammation is often idiopathic; other factors include hypertension, gravitational dependency, medications, strenuous exercise, and infection. Treatments include leg elevation and compression. If there is significant skin inflammation and pruritis, topical steroid and oral antihistamines may be helpful.
Differential diagnosis: Other pigmented purpuric dermatoses (Majocchi, lichen aureus), stasis dermatitis, vasculitis, drug eruption, mycosis fungoides.
Case 2
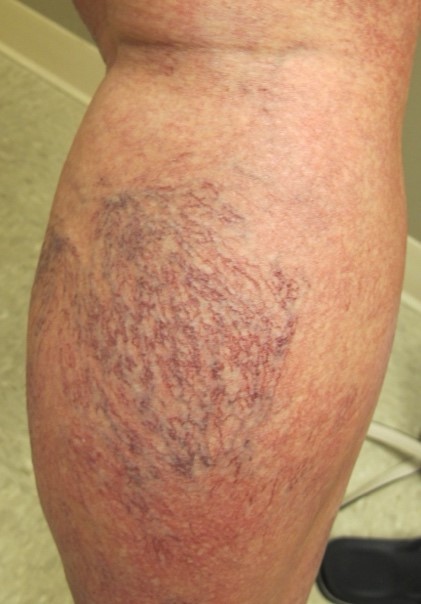
his 45-year-old female presented for treatment of her cosmetically unsightly widespread telangiectasia on the lower extremity. She had cutaneous laser treatment in the past with focal area of clearance. She denied any associated symptoms and her venous duplex ultrasound was negative for reflux.
Diagnosis: Generalized essential telangiectasia
Generalized essential telangiectasia should be considered in the differential diagnosis of a patient who presents with asymptomatic widespread telangiectasia of the lower extremity. Patients often report onset of scattered light-pink telangectasias on the feet, ankles, and distal legs in the 4th or 5th decade of life that become confluent over time. Usually no underlying venous disease or associated actinic damage is apparent. In some cases, there is an autosomal dominant pattern of inheritance. Cosmetic treatment can be successfully performed with sclerotherapy and/or cutaneous laser therapy, but patients should understand that numerous sessions are usually required and recurrence is common. When treating with cutaneous laser, ensure there is slight overlapping of pulses to prevent skipped areas.
Differential diagnosis: spider veins, radiation skin changes, telangiectatic matting due to neovascularization from sclerotherapy/surgery, telangiectasia macularis eruptive perstans (TMEP), ataxia telangiectasia, hereditary hemorrhagic telangiectasia.
Case 3
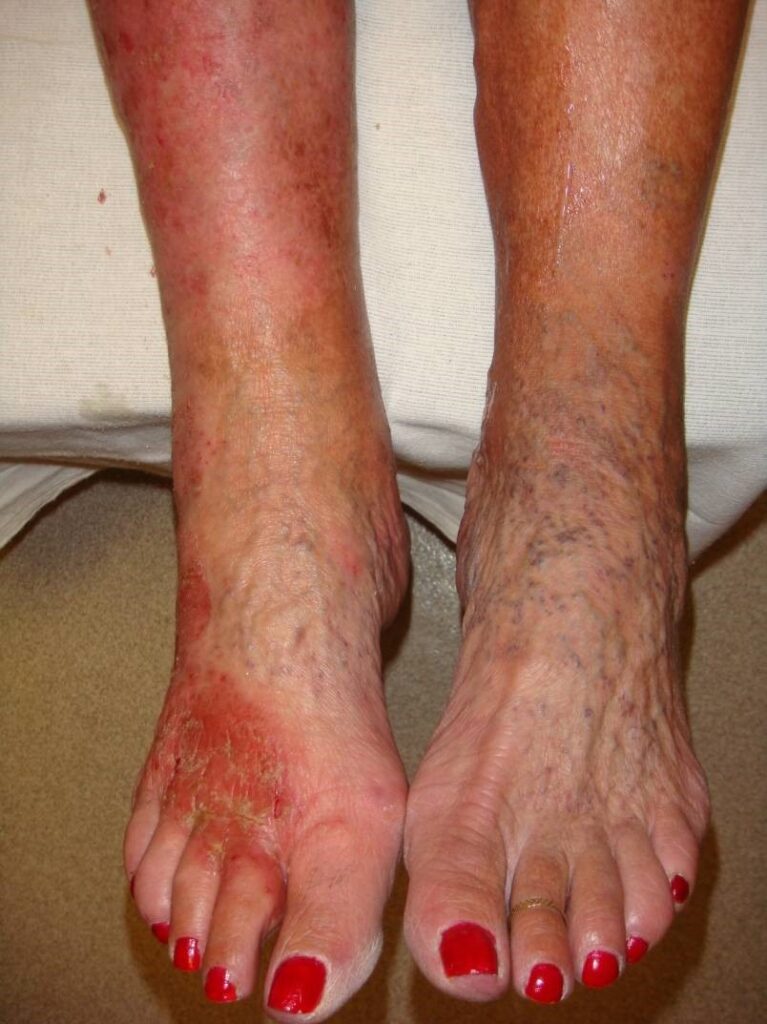
This patient presented with worsening rash to the dorsum of the feet and lateral shins despite daily compression stockings for her chronic venous insufficiency and a trial of high-potency topical steroid and topical antibiotic.
Diagnosis: Allergic contact dermatitis to Neosporin®
Clinicians should consider contact dermatitis when stasis dermatitis worsens or persists despite appropriate high-potency topical steroid therapy. Open wounds and eczematous skin with their compromised skin barriers are highly susceptible to contact sensitization. Patch testing is helpful in determine the specific allergen, which is commonly topical antibiotic (neomycin and bacitracin), topical steroids, and fragrances and lanolin in moisturizers.
Differential diagnosis: Asteatotic dermatitis, atopic dermatitis, secondary bacterial infection (usually some honey crusting), tinea pedis, lichen simplex chronicus (from chronic rubbing), psoriasis.
Case 4
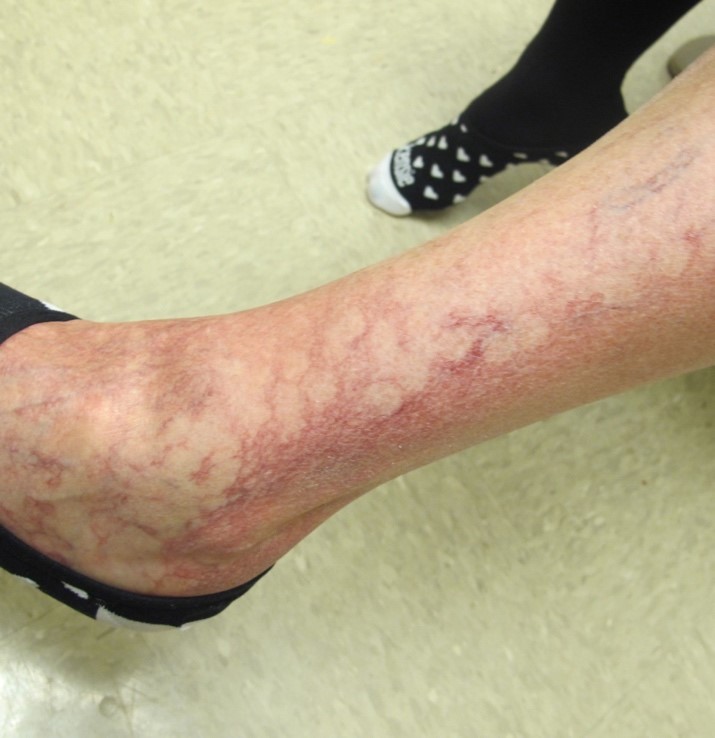
This patient reports new gradual onset of an asymptomatic reticulated hyperpigmented rash to the left lateral and the right medial ankle. She denies any history of ulcerations or rash to the legs and is otherwise healthy. Upon further questioning, she states she has started using a space heater at the corner of her desk.
Diagnosis: Erythema ab igne
Erythema Ab Igne is characterized by reticulated (lace-like) erythema and hyperpigmentation from chronic exposure of the affected area to heat (infrared radiation). This benign condition is due to persistent vascular dilatation from hot water bottle use, space heaters, laptop computers, and heating pads. It usually resolves with cessation of heat exposure, though permanent hyperpigmentation has been reported.
Differential diagnosis: Livedo reticularis (fixed vascular reticulated patterning) associated with numerous systemic diseases including autoimmune disease, vasculitis, antiphospholipid syndrome, cutis marmorata.
Pediatric Thrombosis: Not Just Miniature Adults!

Joann Lohr, MD, FACS, RVT, CWSP
AVF Past President
Chief of Vascular Surgery, William Jennings Bryan Dorn VA Medical Center, Columbia, SC
Pediatric thromboembolism is a dramatically increasing problem with a bimodal distribution affecting neonates and infants and a second peak in adolescence. The highest peak occurs in children <1 month of age. This peak is especially evident in neonates due to advancements in management of their unique illness and invasive supportive care as well as more frequent identification due to improved imaging modalities. The second peak occurs in adolescence with the appearance of acquired DVT, most often in children >11 years of age. Presentation may be very subtle in children compared to adults, with unilateral limb swelling with or without erythema the most common presentation. Subtle changes of cellulitis or unilateral limb pain may occur, but phlegmasia is uncommon. Trending D-dimer values in children with a central venous catheter should raise suspicion of an active thrombotic process especially for values >500 mg/mL. Treatment is anticoagulation with low molecular weight heparin (LMWH) or heparin with targeted anti-Xa levels of 0.3 to 0.7 IU/mL for 3 to 6 months. Subsequent continuation of therapy is determined by the persistence of clinically significant prothrombotic risk factors. Treatment is determined by location and etiology.
LMWH has the advantage of increased DVT efficacy, less intracranial hemorrhage, and a longer half-life than unfractionated heparin. Warfarin is the agent of choice for long term (>3 months) for therapeutic intensity anticoagulation in children. Long-term use of LMWH induces osteoporosis and fractures.
Lower extremity DVT in younger populations is often directly attributed to an inherited thrombophilia, which may result in a missed diagnosis. Recurrent DVT, especially in a younger population, should be evaluated for both thrombophilic and congenital causes. Absence of the inferior vena cava (IVC) may present similarly to thrombophilia with recurrent DVT. Duplex scan of the involved extremity is not adequate to evaluate central anomalies including that of the vena cava. Treatment is conservative for the majority found to have IVC interruption.
Visceral segment DVT is most commonly iliocaval and portal thrombosis. This is commonly seen after liver transplantation. GI bleeding due to variceal congestion or factor consumption or ascites may occur. In children with upper GI bleeding, treatment options include beta blockade, shunting procedures such as transjugular intrahepatic portosystemic shunt (TIPS), or splenectomy. Thrombolytics may be considered.
Short-term sequelae of DVT in the pediatric population include limb edema, pain, discoloration, and pulmonary embolism. Long-term there may be recurrence and post-thrombotic syndrome. Compression stockings are the first line of treatment. Recanalization of chronically thrombosed veins in children has not been studied outside of portal vein thrombosis. The overall mortality in children with DVT is most often associated with their comorbidities.
Neonatal DVT is exceedingly rare, most frequently associated with central venous catheters. Standard LMWH can be used in neonatal DVT with a shortened duration compared to children. For patients with significant risk factors, prophylaxis with antiplatelet agents may be appropriate or LMWH for children <2 months old, 0.75 mg/kg/dose subcutaneously every 12 hours, or for children >2 months old, 0.5 mg/kg/dose every 12 hours subcutaneously. No prophylaxis guidelines have been published for use of IVC filters in children. Direct antithrombin anticoagulant agents for prophylaxis in children is not currently recommended.
Age differences in the hemostatic system of infants and children exist. Adult levels of vitamin K- dependent coagulation factors II, IX, and X and contact factors are not achieved until age 3-6 months. Levels of thrombin inhibitors such as antithrombin and heparin cofactor II are similarly low at birth. They are in the ranges that increase thrombotic risk for heterozygous adults. Levels of alpha-2-macroglobulin are higher in infants and children than in adults. Levels of Protein C and S are low at birth. (Protein S levels approach adult values by age 3 -6 months, but Protein C levels remain low even into childhood). Plasminogen levels are low in newborns and infants. Thrombin generation is decreased (probably because of low prothrombin levels) and delayed in newborns, who have higher risk of bleeding than adults. More study and collaboration are needed, which are currently underway.
References
1. Audu CQ, Wakefield TW, Coleman DM. Pediatric deep venous thrombosis. J Vasc Surg Venous Lymphat Disord. 2019; 7: 452-462.
2. Shoag J, Davis JA, Corrales-Medina FF. Venous thromboembolism in pediatrics. Pediatrics in Review. 2021; 42:78-89.
3. Witmer C, Raffini L. Treatment of venous thromboembolism in pediatric patients. Blood. 2020; 135:335–343.
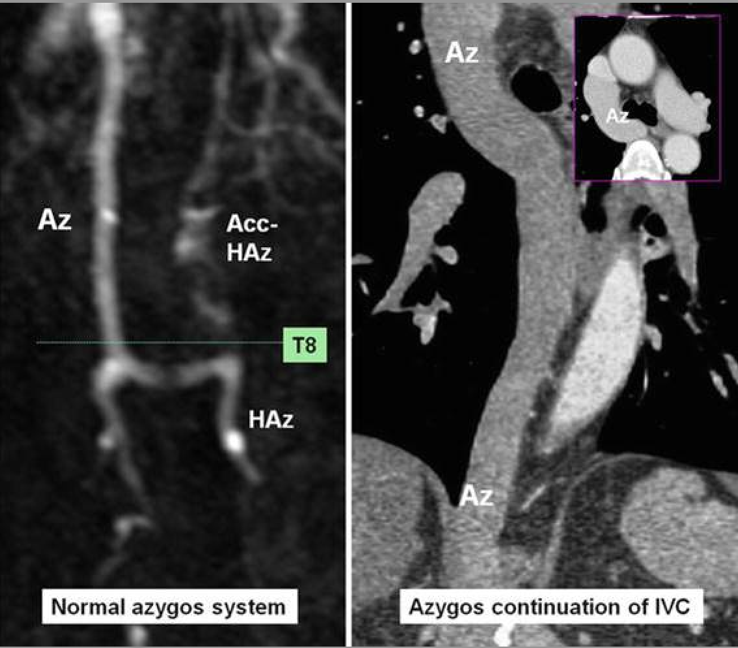
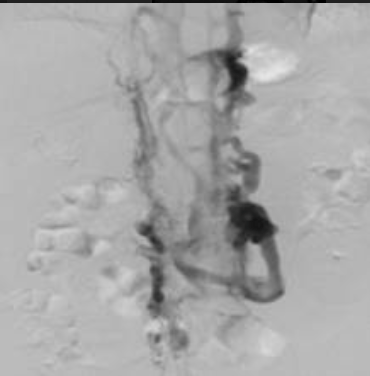
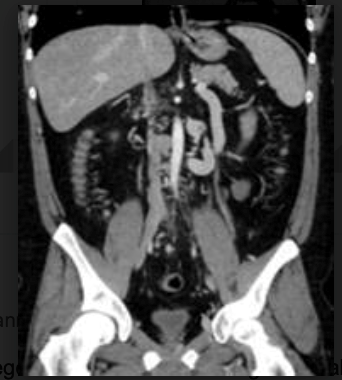
The Underreported Morbidity of Mesenteric Venous Thrombosis

Elizabeth Andraska, MD, MSc
Vascular Surgery Resident, PGY-4
University of Pittsburgh Medical Center

Natalie Sridharan, MD
Member, AVF Newsletter Committee
While MVT is rare in the general population, risk factors include a patient with cirrhosis, cancer, hormonal replacement therapy, or recent laparoscopic abdominal surgery. Many patients with MVT who have cirrhosis will develop portal vein thrombosis with or without thrombosis of the superior mesenteric vein (SMV). In these patients, several series have reported equivalent safety of DOAC and warfarin. Aside from initial therapeutic anticoagulation, little is known or reported on type or duration of anticoagulation. Current practice generally dictates treating it similarly to a lower extremity DVT, with 3e months of anticoagulation if provoked. However, no randomized trials have studied these patients or the outcomes following a course of anticoagulation. Further, the effects of thrombus burden and extension on symptoms, severity, and recurrence are not known. The efficacy or limitations of anticoagulation in these patients should be further described.
Mortality rates following acute MVT are an order of magnitude lower than acute arterial mesenteric ischemia. However, we have previously reported that following acute MVT, most patients will have evidence of portal venous hypertension, mesenteric venous congestion, or recurrent thrombosis on subsequent imaging and nearly half of patients will be admitted within 1 year for abdominal pain. This suggests the presence of a post thrombotic type syndrome of the mesenteric venous system following MVT. Despite the absence of valves in mesenteric veins, the potential exists to develop synechiae and recurrent thrombosis, which could lead to chronic symptoms. While these consequences of MVT may not be fatal, they certainly result in significant morbidity for the patient. With the mainstay of treatment remaining anticoagulation alone, this begs the question: should we be doing more for these patients? Invasive methods to reduce chronic complications of MVT are not well known or described.
Currently, only small case series and case reports describe percutaneous interventions for MVT. In general, these involve either thrombolysis or pharmacochemical techniques to restore flow of the SMV via percutaneous transhepatic, transjugular intrahepatic routes, or indirectly via SMA infusion of thrombolytic agents. Studies reporting these techniques do suggest an improvement in propagation of thrombus and development of portal hypertension but are in general single center with limited patient numbers. These studies are also limited by poor follow-up and incomplete adverse outcomes reporting. The safety of these techniques will need to be better studied and described to determine if this may be an option for these patients.
Mesenteric venous disease remains an understudied and potentially inadequately treated disease. While anticoagulation is a mainstay of treatment, duration and recurrence are not well described. Further, as deep venous interventions continue to evolve, this patient population may benefit from percutaneous interventions.
Reference
Andraska E, Haga L, Reitz K, et al. Acute superior mesenteric venous thrombosis results in high rates of readmission and morbidity. J Vasc Surg Venous Lymphat Disord. 2020; 8:748-755. doi: 10.1016/j.jvsv.2020.01.007. Epub 2020 Mar 3. PMID: 32139329; PMCID: PMC7434641
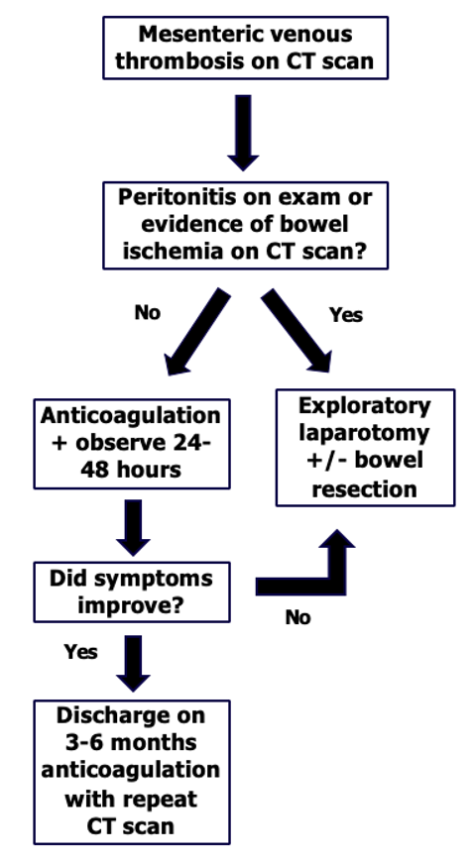
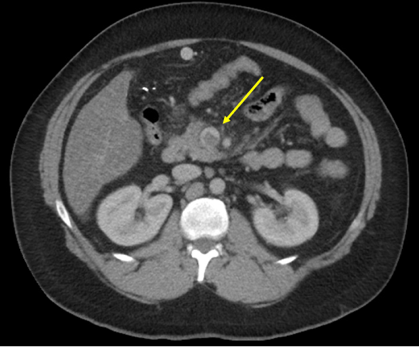
When is a DVT not Just a DVT?

Chandu Vemuri, MD
Past Chair, AVF Guidelines Committee
Principal Investigator, AVF JURY Trial
University of Michigan

Robert Beaulieu, MD, MSE
University of Michigan
Upper extremity swelling in a young patient can be a challenging diagnosis, especially in a healthy patient with no relevant medical history and no indwelling catheters. Subclavian-axillary vein thrombosis may be the culprit; and, while the diagnosis may come quickly, determining the cause and treatment may not be straightforward. The traditional risk factors are often not present. Most importantly, the treatment can differ significantly from lower extremity DVT. Early consultation and referral to a specialist could save the patient significant morbidity and reduce the likelihood of having a lifelong issue.
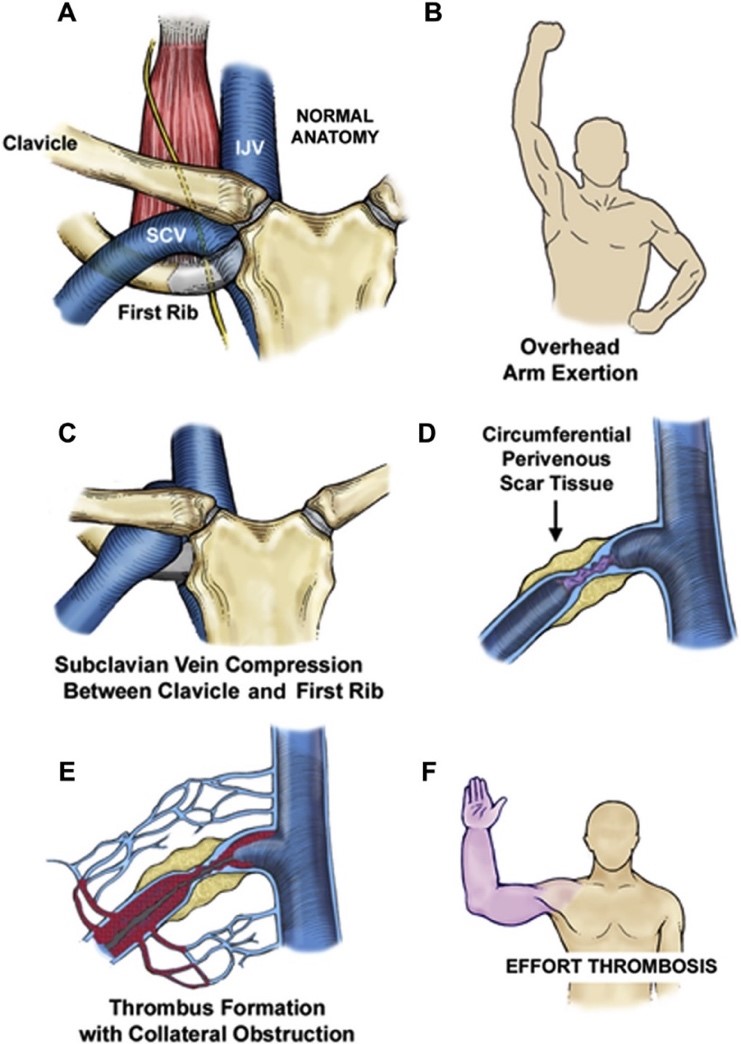
Effort-induced thrombosis (termed venous thoracic outlet syndrome–vTOS) of the axillary and subclavian veins is an important consideration in any young patient presenting with unprovoked upper extremity DVT. Repetitive trauma to the subclavian vein within the thoracic outlet results in stenosis and scarring of the vein which ultimately progresses to thrombosis. Patients may complain of antecedent episodes of limb swelling following repetitive overhead activities. However, the acute thrombosis event can often be sudden and dramatic, presenting with arm swelling, discoloration, and pain. Nearly 40% of patients report complete disability. Duplex sonography is used to secure the diagnosis of upper extremity DVT; yet, in cases of vTOS, this is only one part of the diagnostic workup.
Venography with catheter-directed thrombolysis (CDT) is an important step in both the diagnostic and treatment pathway for many of these patients. The use of CDT within 14 days of symptom onset results in a significantly more rapid return of luminal patency and resolution of symptoms, compared with anticoagulation alone. Further, the dynamic venography performed at the time of lysis allows for visualization of sites of compression and secures the diagnosis of vTOS. The incorporation of intravascular ultrasound can help to determine the need for venous reconstruction at the time of thoracic decompression. Importantly, venoplasty and/or stenting of the subclavian vein should be avoided before surgical decompression of the thoracic outlet as these measures are actually associated with reduced long-term patency, especially with subclavian stenting.
Early surgical decompression of the thoracic outlet through first rib resection should be offered to all patients with vTOS. Anticoagulation alone has been associated with high rethrombosis rates and is not an effective strategy. The term “early” has been a moving target. Initial studies suggested allowing for 3 months between lysis and first rib resection to allow the venous endothelium to heal. However, rethrombosis rates of 16% have been seen during this waiting period, pushing many to advocate for immediate or near-immediate decompression. We typically promote surgical decompression within 3-4 weeks of lytic therapy at our institution. Post-decompression venography, performed 1-3 months after first rib resection, offers an additional measure to treat unresolved stenosis and reports have shown very high technical success rates with >95% patency maintained beyond a year
In many cases of upper extremity DVT, a “DVT” is not “just a DVT.” Only through early recognition and referral to a specialist can we help these patients achieve the best outcomes, reduce recurrence, and allow them to return to a fully functional life.
Reference
1. Vemuri C, Salehi P, Benarroch-Gampel J, McLaughlin L, Thompson R. Diagnosis and treatment of effort-induced thrombosis of axillary subclavian vein due to venous thoracic outlet syndrome. J Vasc Surg Ven Lymph Disord. 2016; 4:485-500.

Publications and Hoofbeats

Ruth L. Bush, MD, JD, MPH
Co-Chair, AVF DEI Ad Hoc Committee
Vice President, AVF Board of Directors
Editor-in-Chief, Journal of Vascular Surgery: Venous and Lymphatic Disorders
One of the goals of the Journal of Vascular Surgery: Venous and Lymphatic Disorders (JVSVL) is to advance the care of patients who have venous and/or lymphatic disease. Another purpose is to explore topics and publish studies that may raise more questions than answers. Science demands inquiry and questioning of norms and existing conclusions. Topics may be considered to be a “zebra” or a “horse” depending on prevalence and incidence. However, perhaps all should be considered provocative subjects deserving of discourse and promoting more primary research and inquiry. For this article, it is not the rare, strange diseases (zebras) that are only worthy of discussion. Rather, it is the hoofbeats we hear every day–the common everyday findings (horses) that warrant more discussion, more basic and clinical research, and less ambiguity. A few examples…
In 2021, Marston et al published a study entitled Early thrombosis after iliac stenting for venous outflow occlusion is related to disease severity and type of anticoagulation (J Vasc Surg Venous Lymphat Disord. 2021; 9:1399-1407). While the title seems fairly straightforward, the conclusions regarding factors associated with early venous re-occlusion including presence of a hypercoagulable state, extent of initial thrombosis, and treatment with low molecular weight heparin for ≥10 days raise suggestions of patient evaluation and consideration of new treatment algorithms. We would encourage more research with larger patient cohorts, perhaps randomized trials looking at post-procedure anticoagulation, and clear metrics for thrombus extent.
Recently, The presence of a pathologic perforator may be predictive of central venous pathology and multilevel disease in severe chronic venous insufficiency by Li et al (J Vasc Surg Venous Lymphat Disord. 2022; Mar;10:402-408) was published. The non-definitive title of this study alone raises questions: “may be predictive?” The title in and of itself begs for more data about refluxing perforators, their contribution to chronic venous insufficiency, and their association with the presence of central venous pathology. Outcomes data are also needed, as well as patient-reported measures such as quality of life.
Bhakta et al published A systematic review and meta-analysis of racial disparities in deep vein thrombosis and pulmonary embolism events in patients hospitalized with coronavirus disease 2019 (J Vasc Surg Venous Lymphat Disord. 2022; 4:939-944.e3). This data aggregate did not find any differences between patients identifying as Black/African or White, though we also know that race is a social construct, not a genetic or biological variable. I can imagine several questions about the categorization of patients into socioeconomic or insurance status, access to care, social supports, etc. The COVID-19 pandemic has opened many other avenues for study including healthcare access, social determinant of health, environmental factors, and the need to study post-pandemic cohorts, long-COVID, delayed healthcare and delayed presentation of disease, and demographic variables including geography.
These are 3 completely different publications, all noteworthy contributions to the literature, but definitely not the final word on these topics. Thus, horses and zebras abound; and we need more primary research and robust studies on varying patient populations to be able to interpret the hoofbeats.
AVF Representation Across the Globe

Dr. Alvaro Orrego with Dr. Luis Rodriguez (El Salvador) at the UIP Congress in Istanbul, Turkey, promoting VENOUS2023.
Swiss Society of Phlebology Highlights: Diversity in Phlebology

Jürg Hafner, Prof., MD
Department of Dermatology, University Hospital, Zurich Switzerland

Dominik Heim, PD, MD
Clinic Hohmad, Thun

Jürg Traber, MD
Director, Venenklinik, Bellevue Kreuzlingen
As Switzerland is a country where different cultures, languages, and characteristics meet, the Swiss Society of Phlebology (SSP) is a conglomerate of several disciplines with the aim to treat the phlebological patient as a whole and at its best.
History
The Swiss Society of Phlebology had its founding scientific meeting in Zurich in 1960. Its first president, Alfred Bolliger (1924-1989), was a charismatic general practitioner and, as a colonel in the Swiss Army, an excellent organizer. In only its second year, the Society held its first international meeting in Agno in Ticino canton in Italian-speaking Switzerland, and used this occasion to establish close, friendly contacts with the French, German, Austrian, and Italian societies of phlebology. Soon afterward, the SSP joined the International Union of Phlebology (UIP). In 1971, the 4th World Congress of Phlebology was held in Lucerne under the leadership of Alfred Bolliger. With 600 participants and 140 papers presented, it was a very large and extremely successful congress by the standards of the time. Such events underlined the importance and prevalence of venous diseases and helped to give phlebology the official recognition it needed as a new medical discipline.
Around the same time, angiology emerged from internal medicine in several European countries. To exploit the synergies among phlebology, angiology, vascular surgery, interventional angiology, and basic vascular research, the 5 independent specialist societies founded the Union of Swiss Societies for Vascular Medicine (USGG).
In 1989, Werner Blättler (Figure 1) became the new SSP president and led the Society for 7 years with great skill and foresight. He conducted research from his phlebology practice on outpatient thrombosis therapy, PTA with stenting of chronic iliac vein occlusion, low-pressure compression therapy, and psychosomatic leg complaints. He was succeeded in 1996 by Albert-Adrien Ramelet (Figure 2) whose scientific work focused on phlebectomy, edema-protective substances, exercise-induced purpura, and bone perforators. Albert-Adrien Ramelet has written a considerable number of phlebology textbooks, sometimes co-authored with other renowned phlebologists. In 2004, Stefan Küpfer (Figures 2 and 4) took over the SSP presidency for 6 years. Stefan Küpfer is an excellent clinical teacher and organizer. He chaired the triennial week-long Venalpina Congress 3 times. Under his leadership, the 3 phlebology diplomas in Switzerland, which ensure a professional approach at the clinic and in practice, were established or updated. Philippe Kern (shown in Figure4) became president of the SSP in 2010. His practice generated important studies on sclerotherapy. He also nurtured the particularly close contact with the French Society of Phlebology. Christina Jeanneret (Figure 3) became SSP president in 2013. Her research focuses on duplex sonography, venous reflux, and wall weakness in varicosis. Under her leadership, serious attention was given to the promotion of female physicians in medical research and in medical committees. Jürg Hafner (seen in Figure 4) has been SSP President since 2019. His scientific field within phlebology is chronic wounds, particularly ulcus hypertonicum Martorell, and research into more patient-friendly medical compression methods. He places great emphasis on the professional growth of young phlebologists. His term of office covers the organization of Venalpina 2020 and 2023. The complete committee is seen in Figure 4.
The important contacts in the international arena were developed in recent years by Albert-Adrien Ramelet during his term as UIP Vice-President. Three years ago, in the summer of 2019, Dominik Heim was able to host the prestigious congress of the European Venous Forum (EVF) in Zurich. Interest was so great that registration had to be closed 2 weeks before the event. It was the “hottest” congress in the history of EVF, not only because of the topics, but the temperature set records for the hottest June for years in Switzerland. Also worthy of note were the 12 medical students, called “Dominik’s Angels,” who were responsible – with the strong team of Anne Taft and Dawn Bond from EVF London–for the smooth running of the congress. And the SSP will bid for the UIP World Congress in 2027…
Health Politics
The SSP is also the political arm of phlebology in Switzerland. In 2020 the SSP published a position paper on the diagnosis, indication, and tailored treatment of truncal varicose veins according to current guidelines, which perfectly matched an earlier position paper on the diagnosis and treatment of reticular and teleangiectasic varicose veins. With these 2 position papers, the SSP upholds the medical nature of major varicose veins and secures reimbursement of the necessary diagnostic and therapeutic measures, whereas treatment of reticular and teleangiectasic varicose veins is classed as aesthetic medicine–which still requires a very high level of expertise but is not covered by health insurance. The SSP also successfully argued for the measurement and distribution of medical stockings by board-certified phlebologists in their medical offices. The agreement on reimbursement for endovenous thermoablation (radiofrequency or laser) of truncal varicose veins was a political milestone that the SSP achieved in 2016.




Membership
Please give a warm welcome to our July & August
New AVF Members
| Candace | Agate | United States |
| Bridget | Bier | United States |
| Sharron | Brownlee | Canada |
| Whitney | Camp | United States |
| Jay | Dema | United States |
| Keetha | Hanlin | United States |
| Tanner | Markus | United States |
| Jonathan | Sadko | Canada |
| Laurie | Dybul | United States |
| Carly | Ferraro | United States |
| Christopher | DeHaven | United States |
| Alexis | Roth | United States |
| Armin | Tabiei | Sweden |
| Eduardo | Leal Villareal | Mexico |
| Manu | Aggarwal | United States |
| Margaret | Arnold | United States |
| Shonda | Banegas | United States |
| Ryan | Messiner | United States |
| Shiffoni | Sukhlal | United States |
| Ali | Ali | United States |
| Sijan | Basnet | United States |
| Brock | Bennett | United States |
| Kathryn | DiLosa | United States |
| Rachel | Grinnan | United States |
| Zachary | Jenner | United States |
| Sooyeon | Kim | United States |
| Patrick | Kim | United States |
| Justin | King | United States |
| Forrest | Linch | United States |
| Turna | Mukherjee | United States |
| Jessica | Rea (Née: Brown) | United States |
| Adam | Reichard | United States |
| Louis | Rucker | United States |
| Nallely | Saldana-Ruiz | United States |
| Kurt | Schultz | United States |
| Jacob | Shapiro | United States |
| Matthew | Tan | United Kingdom |
| Jason | Turner | United States |
| Maday | Gonzalez | United States |